What is the efficacy of standard face masks compared to respirator masks in preventing COVID-type respiratory illnesses in primary care staff?
March 24, 2020

Trish Greenhalgh and Xin Hui Chan, University of Oxford
Kamlesh Khunti, University of Leicester
Quentin Durand-Moreau and Sebastian Straube, University of Alberta, Canada
Declan Devane and Elaine Toomey, Evidence Synthesis Ireland and Cochrane Ireland
Anil Adisesh, University of Toronto, and St. Michael’s Hospital, Toronto, Canada
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
Correspondence to trish.greenhalgh@phc.ox.ac.uk
What is the efficacy of standard face masks compared to respirator masks in preventing COVID-type respiratory illnesses in primary care staff? PDF to Download
Lay Summary by Mandy Payne, Health Watch
UPDATED March 30th 2020
DISCLAIMER: This review was produced at speed in the early weeks of the pandemic. It was based on past research on other diseases, and did not include studies in Covid-19. It has been superseded by other, more extensive reviews which do include studies of Covid-19. There are no plans to update this review.
VERDICT
Standard surgical masks are as effective as respirator masks (e.g. N95, FFP2, FFP3) for preventing infection of healthcare workers in outbreaks of viral respiratory illnesses such as influenza. No head to head trial of these masks in COVID-19 has yet been published, and neither type of mask prevents all infection. Both types of mask need to be used in combination with other PPE measures. Respirator masks are recommended for protection during aerosol generating procedures (AGPs). Rapid reviews on wider PPE measures, and what counts as an AGP, are ongoing.
Most real-world research comparing standard face masks with respirator masks has been in the context of influenza or other relatively benign respiratory conditions and based in hospitals. There are no published head-to-head trials of these interventions in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, COVID-19, and no trials in primary or community care settings. Current guidance is therefore based partly on indirect evidence – notably, from past influenza, SARS and MERS outbreaks – as well as expert opinion and custom and practice.
Policy guidance from various bodies (e.g. Public Health England, WHO) emphasises the need to assess the contagion risk of an encounter and use the recommended combination of equipment for that situation. A respirator mask and other highly effective PPE (eye protection, gloves, long-sleeved gown, used with good donning/doffing technique) are needed to protect against small airborne particles in aerosol-generating procedures (AGPs) such as intubation. For non-AGPs, there is no evidence that respirator masks add value over standard masks when both are used with recommended wider PPE measures.
A recent meta-analysis of standard v respirator (N95 or FFP) masks by the Chinese Cochrane Centre included six RCTs with a total of 9171 participants with influenza-like illnesses (including pandemic strains, seasonal influenza A or B viruses and zoonotic viruses such as avian or swine influenza). There were no statistically significant differences in their efficacy in preventing laboratory-confirmed influenza, laboratory-confirmed respiratory viral infections, laboratory-confirmed respiratory infection and influenza-like illness, but respirators appeared to protect against bacterial colonization.
CONTEXT
Concerns have been raised about the limited personal protective equipment (PPE) provided for UK primary and community care staff with some GP surgeries, pharmacies and care homes having very limited provision. We were asked to find out whether and in what circumstances standard masks are putting healthcare workers at risk of contagion compared to respirator masks. A separate review (ongoing) looks at other aspects of PPE.
BACKGROUND
COVID-19 is spread by four means: contact (direct or via a fomite); droplet infection (droplets from the respiratory tract of an infected individual during coughing or sneezing are transmitted onto a mucosal surface or conjunctiva of a susceptible individual or environmental surfaces); airborne (transmission of infectious agents in small airborne particles, particularly during procedures such as intubation); and faeco-oral.1 2 Coughing and sneezing can generate aerosol particles as well as droplets.
This review considers respiratory protective measures e.g. use of face masks as PPE, to reduce droplet and airborne spread. It should be noted that in one recent laboratory study, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2, the virus that causes COVID-19) survived airborne as long as SARS COV-1 (the virus that causes SARS) when artificially aerosolised and persisted longer on some surfaces.3 This finding is relevant because it suggests that deposited particulates may become resuspended i.e. airborne, when disturbed.
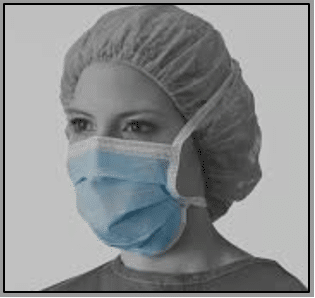
The standard surgical mask (left), also known as a fluid-resistant surgical mask (FRSM), is designed to provide a barrier to splashes and droplets impacting on the wearer’s nose, mouth and respiratory tract. It fits fairly loosely to the user’s face. These single-use masks are used for a variety of procedures in community as well as hospital settings. They should be changed when they become moistened or damaged, and should not be undone and dangled round the neck between procedures. It should be worn with eye protection.
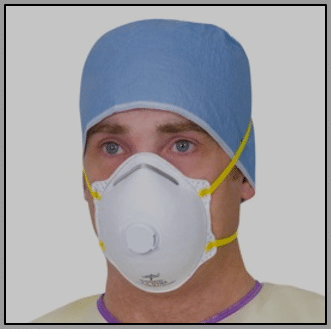
The respirator mask (left), available in the USA as N95 mask and in the UK as an equivalent FFP (‘filtering face piece’) mask, is used to prevent the user from inhaling small airborne particles in aerosol-generating procedures (AGPs). It must fit tightly to the user’s face. There are three categories: FFP1, FFP2 and FFP3. FFP3 provides the highest level of protection. Again, this mask must be worn with eye protection.
Importantly, masks and respirators should not be considered as isolated interventions. Other protection includes hand hygiene, aprons or gowns, goggles or face shields, and gloves.1 4 5 The World Health Organisation has produced technical specifications for these items, based on simulation exercises using data from past SARS and MERS outbreaks.4
A face mask or respirator that is worn without the additional recommended protection will be less effective. Effective training is an essential part of any PPE programme since the correct wearing (donning) and removal (doffing) are key to worker protection. In particular, care should be taken not to contaminate masks on inanimate surfaces.6
CURRENT GUIDANCE
Official UK guidance released in February 2020 claimed that both standard and respirator masks provide 80% protection against SARS-CoV-2 .1 However, this claim referenced a 2017 systematic review that was undertaken before the emergence of SARS-CoV-2 and based largely of trials in seasonal influenza.7 SARS-CoV-2 is known to be both more contagious and more serious than influenza, and may have different patterns of spread. That guidance also recommended the use of heightened protection for AGPs on suspected COVID-19 patients and in all AGP ‘hot spots’ such as intensive care units. It said little about PPE for health care staff in community settings, though it encouraged separation of suspected COVID-19 cases from other patients.
More recently (21st March 2020), Public Health England produced guidance on when to use the different kinds of mask5 and how to put on PPE for non AGP situations.8 These documents emphasise the need to
- Assess the level of risk of infection, especially whether an AGP will be involved (table), before deciding which protection to wear
- Before putting on equipment, perform hand hygiene, remove jewellery, tie hair back and hydrate (feedback from frontline: also go to the lavatory)
- Put on and remove equipment in a way that minimises self-contamination
The figure below, which indicates when to use each type and which procedures are considered ‘aerosol generating’, is taken from PPE guidance.5
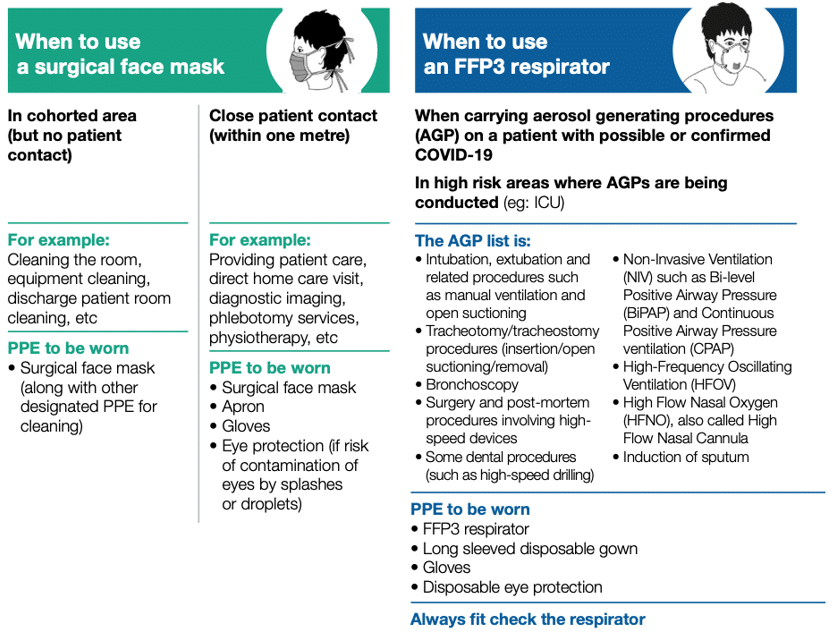
Different PPE for different levels of risk, from PHE guidance5
(‘cohorted area’ is an area where patients with suspected COVID-19 are grouped together)
The WHO distinguishes four situations with three different levels of risk:
- Triage (implicitly, by non-clinical staff without prolonged contact: requires hand hygiene + mask)
- Suspected or confirmed case of COVID-19 requiring healthcare facility admission and no AGPs (hand hygiene, mask, gown, goggles, gloves)
- Suspected or confirmed case of COVID-19 requiring healthcare facility admission and AGP OR collection of specimens for laboratory diagnosis (hand hygiene, respirator, gown, goggles, gloves)
The abovementioned WHO guidance is intended for all healthcare settings but is pitched mainly at secondary care (emergency department or inpatient) settings. A more recent WHO publication specifically considered community and home care settings and offered similar guidance on mask use by healthcare workers (though did not mention triage).9
Primary and community care settings are, by implication, ‘low-risk’ and the guidance does not specifically envisage any situation in which a respirator mask would be needed in primary care. However, the highest number of contacts in the UK will be within primary and community settings including not just general practices but also pharmacies (where many people are attending with symptoms).
It is worth noting that guidance produced by the US Centres for Disease Control recommends respirator masks for both high- and low-risk encounters when patients are suspected of highly contagious and potentially serious conditions such as SARS. However, this guidance was likely based on the precautionary principle and probably did not anticipate the supply shortages currently faced by frontline staff.10
We sought to inform guidance on the use of these different masks in primary care settings.
SEARCH STRATEGY
Starting with two previous systematic reviews known to the authors or their colleagues,11 12 along with a social media search (Twitter) for suggested new papers, we used snowball searching – i.e. seeking later papers on Google Scholar that had cited these references. We identified a very recent meta-analysis done by the Chinese Cochrane Centre, published in early March 2020.13
We supplemented this initial search with a brief database search of Medline and Cochrane databases without date restrictions to identify any additional relevant randomised trials and/or systematic reviews. We used the following key words: “Severe Acute Respiratory Syndrome Virus”, “SARS”, “MERS”, “influenza”, “respiratory tract infections”, “masks”, and the following Mesh term: Influenza, Human/prevention and control.
We limited the set of 126 titles to randomised controlled trials or reviews (12 hits). We repeated a similar search in the Cochrane database of systematic reviews.
CRITICAL APPRAISAL OF KEY STUDIES
All the relevant primary studies had been captured in the Long meta-analysis.13
Based on a brief evaluation of the paper against the AMSTAR II checklist, we judged the review to be of good quality. The authors included six RCTs (five involving healthcare professionals in hospitals and one index patients in the community and household contacts) involving 9171 participants in real-world settings.14-19 They excluded 17 studies (not a trial, not the right intervention, not a real-world trial) and one duplicate trial. Critical appraisal of the included RCTs was done very thoroughly by those authors using risk of bias tools and sensitivity analyses. They commented that some studies had moderate to high risk of bias and only one was community based. Overall, there were no statistically significant differences in preventing laboratory-confirmed influenza (RR = 1.09, 95% CI 0.92 to 1.28), laboratory-confirmed respiratory viral infections (RR = 0.89, 95% CI 0.70 to 1.11), laboratory-confirmed respiratory infection (RR = 0.74, 95% CI 0.42 to 1.29) and influenza-like illness (RR = 0.61, 95% CI 0.33 to 1.14) using N95 respirators and surgical masks. Meta-analysis indicated protective effect of N95 respirators against laboratory-confirmed bacterial colonisation (RR = 0.58, 95% CI 0.43 to 0.78).
AMSTAR II checklist:
- Did the research question include components of PICO? Yes
- Was there a statement that the review methods were established prior to commencement of the review? No
- Did the authors explain the selection of study designs? Yes
- Did the authors use a comprehensive literature search? Yes, three relevant databases searched.
- Did the authors review studies in duplicate? Yes
- Did the authors review data extraction in duplicate? Yes
- Did the authors provide a list of excluded studies and justify exclusions? Yes
- Did the authors describe the included studies in adequate detail? Yes
- Did the authors assess risk of bias satisfactorily? Yes
- Did the authors report on sources of funding of the primary studies? No
- Did the authors use appropriate statistical techniques for meta-analysis? Broadly yes, however decision to use random-effects model seems to have been made on basis of a statistical test for heterogeneity, which we would argue against.
- Did the authors assess the potential impact of risk of bias on the overall results? Yes
- Did they account for risk of bias when interpreting results? Yes
- Did they offer an adequate explanation of heterogeneity? Yes
- Did they investigate for publication bias and discuss its likely impact? No, because of small number of studies available for each pooled estimate (though they had planned to do funnel plots)
CONCLUSION
Trials comparing different kinds of mask have been summarised in a recent high-quality systematic review and provide cautious support for the use of standard surgical masks in non AGPs, though the empirical studies underpinning this conclusion were not in a COVID-19 population, and only one was in a community setting. It is clear from the literature that masks are only one component of a complex intervention which must also include eye protection, gowns, behavioural measures to support proper doffing and donning, and general infection control measures. These wider aspects of PPE will be covered in a further rapid review (ongoing).
Disclaimer: the article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.
REFERENCES
- Official Guidance. COVID-19: Guidance for infection prevention and control in healthcare settings. Version 1.0.: Department of Health and Social Care (DHSC), Public Health Wales (PHW), Public Health Agency (PHA) Northern Ireland, Health Protection Scotland (HPS) and Public Health England 2020. Accessed 21.3.20 https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/874316/Infection_prevention_and_control_guidance_for_pandemic_coronavirus.pd
- Ong SWX, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. Jama 2020
- van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. New England Journal of Medicine 2020
- World Health Organisation. Requirements and technical specifications of personal protective equipment (PPE) for the novel coronavirus (2019-ncov) in healthcare settings. Geneva2020. Accessed 21.3.20 at https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=3&ved=2ahUKEwjS2di4-q3oAhVXh1wKHWeGDvEQFjACegQIARAB&url=https%3A%2F%2Fwww.paho.org%2Fen%2Ffile%2F59297%2Fdownload%3Ftoken%3Dbr2NtqhR&usg=AOvVaw1Sicp3C6m3fjFmMfIeox7R.
- Public Health England. When to use a face mask or FF£ respirator. London2020. Accessed 21.3.20 at https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/874310/PHE_11606_When_to_use_face_mask_or_FFP3_02.pdf.
- Kampf G. Potential role of inanimate surfaces for the spread of coronaviruses and their inactivation with disinfectant agents. Infection Prevention in Practice 2020:100044.
- Offeddu V, Yung CF, Low MSF, et al. Effectiveness of masks and respirators against respiratory infections in healthcare workers: a systematic review and meta-analysis. Clinical Infectious Diseases 2017;65(11):1934-42.
- Public Health England. How to put on personal protective equipment (PPE). London2020. Accessed 21.3.20 at https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/874312/PHE_11606_Putting_on_PPE_02b.pdf.
- World Health Organisation. Advice on the use of masks in the community, during home care, and in health care settings in the context of COVID-19. Geneva: WHO 2020. Accessed 23.3.20 at https://www.who.int/publications-detail/advice-on-the-use-of-masks-in-the-community-during-home-care-and-in-healthcare-settings-in-the-context-of-the-novel-coronavirus-(2019-ncov)-outbreak.
- Chughtai AA, Seale H, MacIntyre CR. Availability, consistency and evidence-base of policies and guidelines on the use of mask and respirator to protect hospital health care workers: a global analysis. BMC research notes 2013;6(1):216.
- Jefferson T, Del Mar CB, Dooley L, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane database of systematic reviews 2011(7)
- Smith JD, MacDougall CC, Johnstone J, et al. Effectiveness of N95 respirators versus surgical masks in protecting health care workers from acute respiratory infection: a systematic review and meta-analysis. Cmaj 2016;188(8):567-74.
- Long Y, Hu T, Liu L, et al. Effectiveness of N95 respirators versus surgical masks against influenza: A systematic review and meta‐analysis. Journal of Evidence‐Based Medicine 2020
- MacIntyre CR, Wang Q, Cauchemez S, et al. A cluster randomized clinical trial comparing fit‐tested and non‐fit‐tested N95 respirators to medical masks to prevent respiratory virus infection in health care workers. Influenza and other respiratory viruses 2011;5(3):170-79.
- MacIntyre CR, Cauchemez S, Dwyer DE, et al. Face mask use and control of respiratory virus transmission in households. Emerging infectious diseases 2009;15(2):233.
- MacIntyre CR, Wang Q, Rahman B, et al. Efficacy of face masks and respirators in preventing upper respiratory tract bacterial colonization and co-infection in hospital healthcare workers. Preventive medicine 2014;62:1-7.
- MacIntyre CR, Wang Q, Seale H, et al. A randomized clinical trial of three options for N95 respirators and medical masks in health workers. American journal of respiratory and critical care medicine 2013;187(9):960-66
- Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers: a randomized trial. Jama 2009;302(17):1865-71.
- Radonovich LJ, Simberkoff MS, Bessesen MT, et al. N95 respirators vs medical masks for preventing influenza among health care personnel: a randomized clinical trial. Jama 2019;322(9):824-33.

