Are there risk factors and preventative interventions for acute respiratory distress syndrome (ARDS) in COVID-19?
June 8, 2020
Catherine Carver and Nicholas Jones
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
University of Edinburgh
Correspondence to c.carver@sms.ed.ac.uk
SHORT ANSWER AND CLINICAL BOTTOM LINE
Seventeen studies were included in this rapid review (1–17), with older age, diabetes, certain blood tests and African American ethnicity amongst the most evidenced risk factors for ARDS. Assessments of risk associated with other comorbidities, symptoms or exposure to medications pre-admission were more inconsistent.
This evidence base should be treated with caution. Most of the studies failed to adjust for key confounders, which is of particular concern to older age, where many comorbidities are known to be more prevalent. This, combined with generally small sample sizes, leaves the results open to significant risk of bias.
Several factors may affect the generalisability of the results of these studies. In particular, the definition of COVID-19 cases varied across studies and whilst several studies used the Berlin(18) or WHO interim guidance(19) definitions of ARDS, others were not explicit.
In short, we found a lack of robust data regarding risk factors or prevention of COVID-19 ARDS and identified an urgent need for high quality research in this area.
BACKGROUND
Acute respiratory distress syndrome (ARDS) is a type of “acute diffuse, inflammatory lung injury”(18) that can be caused by an array of events, from bacterial or viral infection, to blood transfusion or near drowning (20). It has been identified as a serious and frequently lethal complication of COVID-19, with Wu reporting a mortality rate of 52.4% amongst COVID-19 ARDS patients (3). Symptoms include breathlessness, dizziness and rapid heart rate (21). It is typically defined using the 2012 Berlin criteria (18) or the WHO interim guidance (19), both based on a constellation of factors including timing of symptoms, oxygenation and chest imaging. The rate of ARDS in COVID-19 varies by the cohort being reported, but appears to be high among hospitalised cohorts. A recent meta-analysis which included 50,466 COVID-19 cases described an ARDS incidence of 14.8% (95% CI: 4.6-29.6)(22).
Given the potential severity of ARDS, being alert to which patients are at an increased risk of ARDS or being aware of interventions to reduce the likelihood of ARDS has the potential to be influential in clinical practice. It has been suggested COVID-19 ARDS has unique features (23), which may mean the existing evidence on prognostic factors for ARDS is not directly applicable. For this reason, this review focuses on COVID-19 specific knowledge and aims to determine if there are any known risk factors for ARDS in this infection and whether there are any interventions to prevent ARDS developing.
SEARCH STRATEGY
Search terms: The CDC suggested COVID-19 search terms were combined with relevant text words and MeSH terms for risk or prevention (including risk*, predict*, prevent*, probab*, control and associat*) and for ARDS (including ARDS, acute respiratory distress syndrome, critical illness, critical care and acute lung injury).
Databases: PubMed, Embase, MedRxiv. Searches limited to 2020 and are current to May 17th 2020.
Hits: 1157 unique citations
Additionally, hand sifted citations of the paper by Wu et al(3) were included as this was identified as the most cited paper and their primary aim was identifying ARDS risk factors.
Included papers: 17 (Excluded studies with 10 or fewer total ARDS cases because of high risk of bias. Two systematic reviews with meta-analyses were excluded due to significant concerns regarding their methodology (24,25))
CRITICAL APPRAISAL CHECKLIST
CASP or Joanna Briggs Institute checklists, depending on study design, were used by both review authors to independently appraise all papers. The papers were generally found to be of low quality, primarily because they had small overall sample sizes, with smaller numbers of patients who developed ARDS. In addition, most papers did not adjust for covariates in the relationship with ARDS and no study adjusted for all key confounders. These problems were compounded by the fact inclusion criteria often had to be presumed rather than being explicit, the use of differing definitions of COVID-19 cases and the fact the definition of ARDS and some comorbidities were varied and not always specified, rendering the data too heterogenous to conduct a meta-analysis. Moreover, most studies were retrospective cohort studies dependant on an examination of electronic medical records and therefore dependent on the completeness of those records.
STUDY CHARACTERISTICS
Sixteen of the studies described hospitalised COVID-19 patients, leaving Khan et al (10) as the only paper that included medical records from a mix of inpatient, outpatient, and specialty care services. In the case of Yu et al (15), hospitalisation was not necessarily a marker of disease severity because all outpatients were required to be quarantined in the hospital if infection with 2019-nCoV was confirmed. In Lian et al (6) all PCR confirmed COVID-19 cases in Zhejiang Province were included, but the paper implies all were hospitalised but perhaps due to public health requirements rather than disease severity: “all patients were allocated at specific hospitals for unified treatment according to the government emergency rule”. Such public health rules may have influenced admission in other studies, but these two stated these factors explicitly.
With regards COVID-19 case definition, 14 of the papers used PCR, ‘lab confirmed’ (likely to be PCR) or WHO interim guidance definitions. Two were unclear as to their COVID-19 case definition (5,16), whilst Khan included a variety of codes from electronic medical records, including “COVID-19, virus identified” but also others such as “Coronavirus infection, unspecified”.
A selection of ARDS definitions were included, as outlined below in Table 1.
Table 1. ARDS definition used in each included study
| Study |
ARDS definition |
| Shi 2020a (1) |
Berlin |
| Li 2020 (2) |
Berlin |
| Wu 2020 (3) |
WHO interim guidance |
| Dreher 2020 (4) |
Berlin |
| Bi 2020 (5) |
Not stated |
| Lian 2020 (6) |
Not stated |
| Liu 2020 (7) |
Berlin |
| Fadel 2020 (8) |
Berlin |
| De la Rica 2020 (9) |
Not stated |
| Khan 2020 (10) |
Not stated |
| Zhang 2020 (11) |
WHO interim guidance |
| Palaiodimos 2020 (12) |
Not stated |
| Shi 2020b (13) |
Berlin |
| Mahevas 2020 (14) |
Unclear |
| Yu 2020 (15) |
Bespoke: acute-onset oxygenation index (arterial partial pressure of oxygen/fraction of inspired oxygen) ≤ 300 mm Hg and a chest radiograph showing patchy shadows |
| Zhu 2020 (16) |
WHO interim guidance |
| Inciardi 2020 (17) |
Not stated |
SUMMARY OF FINDINGS FROM MOST USEFUL PAPERS
DEMOGRAPHICS
Older age
Four studies reported older age was associated with an increased risk of ARDS. In a retrospective cohort study of 201 hospitalised patients with confirmed COVID-19 pneumonia(3), 84 (41.8%) developed ARDS. Wu(3) noted that the median age of ARDS patients was 58.5 years, compared with 48 years for non-ARDS patients. They calculated being aged 65 years or over was associated with a 3.26 increased risk of ARDS (95% CI 2.08-5.11 p<0.001) compared to the under 65s. They also found that patients who developed ARDS and were aged 65 years or over had a 6.17 increased risk of death (95% CI, 3.26-11.67; P<0.001) compared to ARDS patients under 65.
Lian et al (6) also found older age to be a risk factor for ARDS in COVID-19. They reported 5.37% (35/652) of those aged under 60 developed ARDS, compared to 16.91% (23/136) of 60 and overs (p <0.001). Similarly, in a non-peer reviewed preprint Liu and colleagues(7) noted that the median age of ARDS patients (n=53) was 61 (IQR 52-70), compared to 49 (IQR 37-59) for non-ARDS patients (n=56) (p<0.001). These medians are similar to those of Wu(3). Yu also reported a statistically significant relationship between ARDS and age on univariate analysis, but the effect was very small and was no longer significant on multivariate analysis(15).
Supporting data on the link between ARDS and older age can be found in a non-peer reviewed preprint by Bi et al (5) that describes a cohort of 420 COVID-19 patients from Shenzen, China (39 ARDS cases). Instead of looking at risk of developing ARDS, they examined predictors for faster clinical progression to ARDS. When comparing the rate of progression to ARDS, they found being age 60 or over was associated with a 21.9 increased risk compared to 0-39 year olds. However, the 95% confidence interval on this ratio was exceedingly wide (5.2-91.7), demonstrating the imprecision of the result.
Contrastingly, two smaller studies (4,9), each with approximately 20 ARDS cases, did not report a significant relationship between age and ARDS. However, they may not have had the sample size or population variation to demonstrate differential risk. A third low quality study with 45 ARDS cases also found no significant relationship (12), but the generalisability of its findings to the average UK hospital were limited by the fact it was a tertiary care centre (potentially implying a particularly high risk group) and 50% of patients were African American.
Gender
Of interest was the finding that whilst COVID-19 mortality has been linked to being male(26), none of the papers we included reported a significant difference in risk of ARDS between men and women(3–5,7,9).
Ethnicity
In a preprint of a large study of electronic medical records in the United States, Khan et al(10) concluded that African Americans were at a higher risk of ARDS than White individuals. After propensity matching of cohorts was performed accounting for age, gender, BMI, and major comorbidities they reported that 7.89% (147/1862) of African Americans with COVID-19 developed ARDS, compared to 4.03% (75/1862) of White individuals, giving an increased risk of 1.96 times (1.49,2.56; p< 0.0001). This is in line with recent research suggesting higher mortality in black individuals, even after adjusting for various covariates(26). Overall, this was a fair quality study, but important limitations included challenges around accurate coding of COVID-19, a lack of a definition of ARDS and the absence of accounting for deprivation. The latter is a potentially important confounder given its potential implication in COVID-19 (26), its link to accessing care in the American system and the inequalities between African American and White populations (27).
COMORBIDITIES
Several studies examined links between comorbidities and ARDS risk, with mixed results.
Hypertension
Hypertension was associated with an increased risk of ARDS in two low quality studies. Wu et al(3) calculated that hypertension was associated with an unadjusted hazard ratio of 1.82 for developing ARDS (95% CI, 1.13-2.95, p=0.01). Supporting this data was a paper by Zhang et al (11), which found hypertension carried an adjusted hazard ratio of ARDS of 1.61 (1.32,1.97; p0.000). The adjusted variable included age, gender, comorbid diabetes, cerebrovascular diseases, and chronic renal disease. The ARDS sample size of Zhang was unclear, but as the management of 302 patients involved either non-invasive or invasive ventilation, the ARDS population is likely to be larger than most of the studies we found.
Running against the findings of Wu and Zhang were three smaller low-quality studies. Dreher(4) found no significant increased risk of ARDS associated with arterial hypertension (non-ARDS: 73% 19/26 hypertension vs ARDS 67% 16/24 hypertension.) Similarly, de la Rica (9) found no relationship with pre-existing hypertension (non-ARDS: 82% 22/27 ARDS: 55% 11/20) p0.101). These higher hypertension rates than in Wu across all patients may relate to the different median age of patients (older in Dreher and de la Rica) and potentially differing hypertension prevalence and diagnostic rates between China (Wu) and Europe (Dreher, de la Rica).
As with age, Bi et al(5) offered a hazard ratio that compared rate of progression to ARDS, rather than just risk of developing ARDS. They calculated the hazard ratio of hypertension was 3.6 (95% CI, 1.8-6.9, no p-value).
Diabetes (type not specified by most studies)
Wu(3) reported a statistically significant relationship between diabetes and ARDS, with 5.1% (6/117) of non-ARDS cases having diabetes vs 19% of ARDS cases (16/84) (p=0.002). They calculated that diabetes carried a 2.34 increased risk of ARDS (95% CI, 1.35-4.05, p=0.002). Liu(7) supported this, with 1.8% (1/56) of non-ARDS having diabetes vs 20.8% (11/53) of ARDS cases (p=0.002). Huang (which we did not include as a core paper due to having already included the data their review was based on) used this data to construct a meta-analysis which found diabetes was associated with relative risk of ARDS of 4.64 (95% CI 1.86, 11.58, p = 0.001)(28). Further support for this relationship comes from Shi 2020b, which found ARDS was more common in diabetic patients (ARDS rates: non-diabetics 17/153 11.1% vs diabetics 38/153 24.8% p0.002)(13).
However, contrasting with this was the result of Dreher et al(4), who reported a non-significant finding of diabetes in 54% of non-ARDS cases (14/26) vs 63% of ARDS cases (15/24). This is a very high rate of diabetes, both compared to Wu and to the over 60s population of Germany at large (29), which raises questions about the generalisability of data from Dreher’s patients. Mirroring Dreher, de la Rica found no significant relationship between type 2 diabetes and ARDS, but had just 14 diabetics in total in their study(9).
Bi’s(5) assessment of rate of progression to ARDS, rather than just risk of developing ARDS, assigned diabetes a hazard ratio of 5.5 (95% CI, 2.7-11.3, no p-value).
Cardiac disease
Inciardi et al(17) examined the relationship between ARDS and cardiac disease (using a broad definition that included atrial fibrillation, heart failure and coronary artery disease) in 99 consecutive patients admitted to hospital with COVID-19 pneumonia. They found no significant difference in ARDS cases between the cardiac patients and non-cardiac patients, but only had 19 cases of ARDS total in their sample and the study is likely underpowered.
Respiratory disease
Dreher(4) reported “pre-existing respiratory disease” as having a significant association with ARDS (42% non-ARDS had this vs 58% of ARDS (No p-value, but described as significant). However, this was a broad grouping that covered COPD, obstructive sleep apnoea syndrome, bronchial asthma and ‘other pulmonary diseases’. Looking at another composite definition, Bi offered a hazard ratio that compares rate of progression to ARDS for “chronic lung disease”. They reported a hazard ratio of 4.2 (95% CI, 1.5-11.8, no p-value)(5).
High BMI
Three studies examined the impact of BMI on ARDS risk with inconsistent results. In a study that included 24 ARDS cases, Dreher (4) found 23% of the 26 non-ARDS cases had a BMI of 30 or greater, vs 46% of ARDS cases (p-value not provided, but described as significant). On the other hand, examining 46 ARDS cases and using different BMI groupings from Dreher (BMI < 25, BMI 25–34, and BMI ≥ 35) Palaiodimos (12) found no significant relationship between BMI and ARDS. Similarly, Yu found no significant relationship between BMI and ARDS in a study that included 24 ARDS cases (mean BMI in non-ARDS patients 22.64 vs 24.26 in ARDS cases, p0.053) (15). All three were low quality studies and neither Dreher nor Palaiodimos conducted any adjustment for confounding.
Other comorbidities all with non-significant results
Several other comorbidities were investigated, particularly by Dreher(4), but with no statistically significant risk difference between ARDS and non-ARDS. Dreher examined a multitude of comorbidities including cancer, chronic kidney failure, chronic hepatitis and chronic liver failure, but the sample size per comorbidity was under 10 and therefore underpowered to detect differences.
ACUTE ILLNESS
Two studies examined acute illness and ARDS in COVID-19. The first, by Li and colleagues, looked at whether patients who were severely ill with COVID-19 were more likely to have ARDS, and found there was a correlation(2). Of those with nonsevere COVID-19, 27 of 279 (9.7%) had ARDS as an outcome. This compares with 183 of 269 (68.0%) of severe COVID-19 patients having ARDS (p=.000). Whilst this has prima facie believability, this finding is problematic because ARDS could well be severe disease, so the outcome and exposure may be the same thing. This is ameliorated by the fact disease severity was assessed on admission, and ARDS may have developed later, but it is unclear when most ARDS diagnoses were made. With regards initial severity, Liu(7) also found that having a lower PaO2:FiO2 on admission was associated with ARDS as an outcome (475mm Hg (398-524) vs 145 mm Hg (108-250) p<0.001). Of course, as a low PaO2:FiO2 is part of the definition of ARDS this again may just reflect that patients already had ARDS.
Shi et al(1), also looked at an acute event – cardiac injury, defined as 99th-percentile upper reference limit high-sensitivity troponin I (hs-TNI). They found ARDS was more common in patients with cardiac injury (58.5%, 48/82 with cardiac injury had ARDS vs 14.7%, 49/334 without cardiac injury had ARDS; P < .001). One significant concern with this study is that they started with a cohort of 1004 patients, with exclusions for various reasons including 229 for missing cardiac biomarkers. These 229 patients were notably different from the rest of the cohort, for instance they were more likely to be younger and to be female, so had they been tested and their results included, the ARDS risk associated with cardiac injury may have been different.
Cardiac injury as a risk factor for ARDS was mentioned in a meta-analysis by Santoso et al, however we have not included their data because it was based on 2 studies – this one by Shi, and another that reported a composite outcome of ARDS, sepsis and respiratory failure. Based on this composite outcome we question the validity of relying on this data for an ARDS specific meta-analysis and therefore have not reported it.
LABORATORY RESULTS
Five studies looked at lab results and ARDS. Wu(3) alone reported on over 30 lab results, several of which were significant but there were too many to describe here – for a full understanding please refer to the original manuscript. Instead, we will focus on those results that were found to be statistically significant by both Wu(3) and Liu(7), both of which had more than 50 ARDS cases. It is important to note that different studies had different results, so higher/lower describes the relationship as reported between ARDS and non-ARDS patients within each study. Where available, the most stringent adjusted results were used to define whether a result was a significant finding by a paper.
Table 2. Risk of ARDS in relation to relative laboratory values
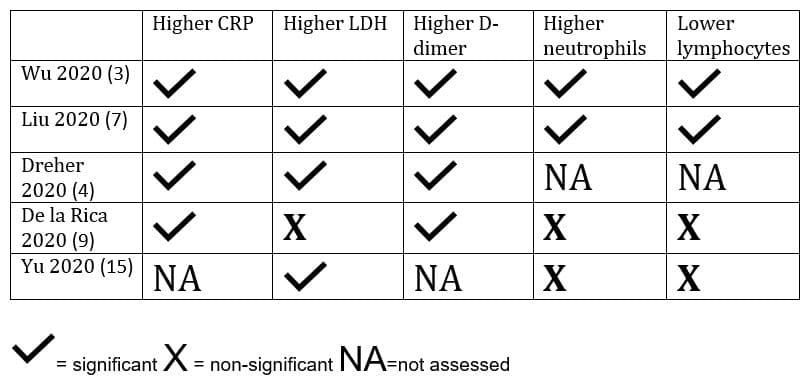
Wu reported bivariate Cox regression for these five initial test results, with the following statistically significant hazard ratios for ARDS reported:= significant X = non-significant NA=not assessed
Table 3. Reported hazard ratio for risk developing ARDS in relation to select laboratory results
|
Hazard ratio |
95% CI |
p-value |
| Higher LDH |
1.61 |
1.44-1.79 |
<0.001 |
| Higher D-dimer |
1.03 |
1.01-1.04 |
<0.001 |
| Higher hs-CRP |
4.81 |
1.52-15.27 |
0.008 |
| Higher neutrophils |
1.14 |
1.09-1.19 |
<0.001 |
| Higher lymphocytes |
0.37 |
0.21-0.63 |
<0.001 |
Whilst significant, these results often had wide spreads, with the interquartile ranges of ARDS and non-ARDS patients overlapping for the five described tests in both Wu(3) and Liu(7).
SYMPTOMS
A range of different symptoms were reported across four papers. For instance, Dreher(4) alone described 13 different symptoms, with 10 having a total n<10. As this is a rapid review, we have restricted reporting to those highlighted by Wu(3), which had the largest sample of ARDS patients, which had a total n of 50 or above for each of the described symptoms.
Fever
Wu(3) reported that fever of 39C or above was associated with a 1.77 times increased risk of ARDS (95% CI 1.11-2.84, p=.02). One potential flaw in Wu’s measurement is that they relied upon self-reported highest temperature prior to hospital admission, which may have been inaccurate based upon who took it and how, as well as being subject to recall bias. Neither Dreher(4), Liu(7) or Bi(5) described a significant relationship between initial fever and ARDS. Although 92% of ARDS vs 72% of non-ARDS had initial fever in Dreher, no p-value is offered and unlike other findings it is not described as significant in the text.
Dyspnoea
Wu(3) also reported initial dyspnoea was statistically more common in ARDS patients (59.5%, 50/84 vs 25.6% 30/117 p<0.001). Dreher(4) also examined dyspnoea, whilst they do not report on whether the result is significant, they reported 16 ARDS patients (64%) had initial dyspnoea compared with 8 (32%) of non-ARDS patients. Bi(5) found a statistically significant impact on rate of progression to ARDS between those with dyspnoea and those without: hazard ratio 7.2 (95% CI 3.2-16.0).
A range of other symptoms were found to have non-significant relationship with ARDS by Wu(3), including cough (also non-significant in Dreher(4), Liu (dry cough)(7) and Bi(5)), productive cough (not examined elsewhere), and ‘fatigue or myalgia’. Fatigue was examined in other studies, but the studies conflicted – whereas initial fatigue was only found amongst non-ARDS patients by Dreher (n=6), it was statistically significantly more common as an entering complaint in ARDS patients in Liu (67.9%, 36/53 vs 46.4%, 26/56, p0.02).
INTERVENTIONS
None of the studies we identified were designed to robustly assess the preventative powers of interventions against ARDS. For instance, the statistically significant differences in oxygen therapy during hospitalisation in Wu are likely a reflection of severity of illness – it doesn’t provide any meaningful insight into the preventative powers of oxygen therapy. Non-invasive mechanical ventilation is used to treat ARDS, so we’re probably simply seeing that people with ARDS are treated for ARDS.
Some studies examined correlations between medications (either before or after admission) and ARDS. Dreher(4) found no significant relationship with pre-existing medications (such as ARBs or ACE inhibitors) but had relatively small sample sizes for each medication (range of n=4-23). Zhang(11) examined hypertensive patients and found no significant relationship between ARDS and use of ACE inhibitors or ARBs (HR 0.65, 95% CI0.41-1.04 p0.07) after propensity matching for a broad range of key covariates. The ARDS sample size was not stated in this paper, but ~300 patients received NIV or invasive ventilation so it likely represents a reasonable ARDS cohort.
Liu(7) found no statistically significant association between inpatient or fever clinic treatments, such as antibiotics or antivirals, and ARDS. In the case of antivirals and antibiotics this may have been because 96.3% of the sample were prescribed these drugs, making it difficult to detect differences between the groups.
Wu(3) found that ARDS patients were more likely to be treated with methylprednisolone during their admission and less likely to have been given antivirals. However, these results may well just reflect a different clinical course for these patients, rather than informing us about whether medications represent risk factors.
Mahevas(14) conducted a comparative observational study based on routine data to assess hydroxycholoroquine use against several outcomes, including ARDS prevention. The rate of survival without acute respiratory distress syndrome was 70% in the treatment group and 74% in the control group (1.2, 0.7 to 2.2), a non-significant finding from a low-quality study with 48 ARDS cases.
In a multi-centre, quasi-experimental design study, Fadel(8) found a significant relationship between early short course corticosteroids and ARDS (ARDS Pre-protocol 31/81 38.3%, ARDS post-protocol 33/132 26.6% p0.040). We had several concerns with regards this study, for instance over half of those in pre-protocol group still received steroids, but tended to start these significantly later. Moreover, the post-protocol group seem to have been more unwell – more chronic kidney disease, heart failure, hypertension, more with cough, fever and myalgia.
DISCUSSION
There remains significant scope and need for studies to robustly determine risk factors and preventative interventions for ARDS in COVID-19. The seventeen studies we identified generally suffered from small sample sizes and a lack of adjustment for confounders. However, their results do perhaps provide inspiration for the types of risk factors to be examined in more depth. Older age was one of the most consistently reported risk factors, however, as mentioned, the studies did not adjust for confounders and we know many comorbidities are more common in older age, including diabetes and hypertension. Diabetes specifically was identified as a risk factor for ARDS by three studies. The results for other comorbidities, symptoms and previous drug therapy were more mixed.
Of interest was the finding that whilst severity has been linked to being male, the papers we identified did not find a significant relationship between being male and ARDS. However, in keeping with findings about mortality, African American ethnicity was identified as a risk factor for ARDS in a preprint describing a large study of fair quality after adjustment for several confounding factors.
LDH, CRP, D-dimer and neutrophils were found to be statistically significantly higher in those with ARDS as an outcome, whilst lymphocyte counts were lower in ARDS patients. Given the hypothesis that COVID-19 ARDS results from a cytokine storm(30–32), better understanding the predictive value of these lab results may also guide possible preventative interventions.
Whilst we found papers in the literature which hypothesised potential interventions to prevent ARDS, such as valproate (33) or multiple sclerosis drugs (34), none of the studies we included had a robust methodology for assessing interventions for the prevention of ARDS. One pre/post study (8) of the introduction of an early short course of corticosteroids, finding a statistically significant ARDS free survival in the control group. However, this study had several major limitations, for instance over half the pre-protocol group also received steroids just later in their admission.
Several factors may affect the generalisability of the results of these studies. In particular, the definition of COVID-19 cases varied across studies and whilst several studies used the Berlin or WHO interim guidance definitions of ARDS, others were not explicit and one used a bespoke definition.
Disclaimer: the article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.
AUTHORS
- Catherine Carver holds a Chancellor’s Fellow PhD studentship at the University of Edinburgh’s Usher Institute. She has a medical degree from the University of Aberdeen and a Masters in Public Health from Harvard.
- Nick Jones is a GP and Wellcome Trust Doctoral Research Fellow working at the University of Oxford Nuffield Department of Primary Care Health Sciences
REFERENCES
- Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020 Mar 25;
- Li X, Xu S, Yu M, Wang K, Tao Y, Zhou Y, et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol. 2020 Apr 12;
- Wu C, Chen X, Cai Y, Zhou X, Xu S, Huang H, et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. 2020;
- Dreher M, Kersten A, Bickenbach J, Balfanz P, Hartmann B, Cornelissen C, et al. The Characteristics of 50 Hospitalized COVID-19 Patients With and Without ARDS (17.04.2020). Dtsch Ärztebl Int. 2020;(117):271–8.
- Bi Q, Hong C, Meng J, Wu Z, Zhou P, Ye C, et al. Characterizing clinical progression of COVID-19 among patients in Shenzhen, China: an observational cohort study. medRxiv. 2020 May 13;2020.04.22.20076190.
- Lian J, Jin X, Hao S, Cai H, Zhang S, Zheng L, et al. Analysis of Epidemiological and Clinical Features in Older Patients With Coronavirus Disease 2019 (COVID-19) Outside Wuhan. Clin Infect Dis [Internet]. [cited 2020 May 5]; Available from: http://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciaa242/5811557
- Liu Y, Sun W, Li J, Chen L, Wang Y, Zhang L, et al. Clinical features and progression of acute respiratory distress syndrome in coronavirus disease 2019. medRxiv. 2020 Feb 27;2020.02.17.20024166.
- Fadel R, Morrison A, Vahia A, Smith ZR, Chaudhry Z, Bhargava P, et al. Early Short Course Corticosteroids in Hospitalized Patients with COVID-19. medRxiv. 2020 May 5;2020.05.04.20074609.
- Rica R de la, Borges M, Aranda M, Castillo A del, Socias A, Payeras A, et al. Low albumin levels are associated with poorer outcomes in a case series of COVID-19 patients in Spain: a retrospective cohort study. medRxiv. 2020 May 11;2020.05.07.20094987.
- Khan A, Chatterjee A, Singh S. Comorbidities and Disparities in Outcomes of COVID-19 Among African American and White Patients. medRxiv. 2020 May 15;2020.05.10.20090167.
- Zhang Peng, Zhu LiHua, Cai Jingjing, Lei Fang, Qin Juan-Juan, Xie Jing, et al. Association of Inpatient Use of Angiotensin Converting Enzyme Inhibitors and Angiotensin II Receptor Blockers with Mortality Among Patients With Hypertension Hospitalized With COVID-19. Circ Res [Internet]. [cited 2020 May 19];0(0). Available from: https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.120.317134
- Palaiodimos L, Kokkinidis DG, Li W, Karamanis D, Ognibene J, Arora S, et al. Severe obesity is associated with higher in-hospital mortality in a cohort of patients with COVID-19 in the Bronx, New York. medRxiv. 2020 May 9;2020.05.05.20091983.
- Shi Q, Zhang X, Jiang F, Zhang X, Hu N, Bimu C, et al. Clinical Characteristics and Risk Factors for Mortality of COVID-19 Patients With Diabetes in Wuhan, China: A Two-Center, Retrospective Study. Diabetes Care. 2020 May 14;
- Mahévas M, Tran V-T, Roumier M, Chabrol A, Paule R, Guillaud C, et al. Clinical efficacy of hydroxychloroquine in patients with covid-19 pneumonia who require oxygen: observational comparative study using routine care data. BMJ. 2020 May 14;369:m1844.
- Yu T., Cai S., Zheng Z., Cai X., Liu Y., Yin S., et al. Association Between Clinical Manifestations and Prognosis in Patients with COVID-19. Clin Ther [Internet]. 2020;((Yu, Cai, Peng, Xu) Department of Infectious Diseases, Nanfang Hospital, Southern Medical University, Guangzhou, Guangdong Province, China). Available from: http://www.elsevier.com/locate/clinthera
- Zhu L, She Z-G, Cheng X, Qin J-J, Zhang X-J, Cai J, et al. Association of Blood Glucose Control and Outcomes in Patients with COVID-19 and Pre-existing Type 2 Diabetes. Cell Metab. 2020 May 1;
- Inciardi RM, Adamo M, Lupi L, Cani DS, Di Pasquale M, Tomasoni D, et al. Characteristics and outcomes of patients hospitalized for COVID-19 and cardiac disease in Northern Italy. Eur Heart J. 2020 May 14;41(19):1821–9.
- Acute Respiratory Distress Syndrome: The Berlin Definition. JAMA. 2012 Jun 20;307(23):2526–33.
- WHO. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected [Internet]. 2020 Mar. Available from: https://www.who.int/docs/default-source/coronaviruse/clinical-management-of-novel-cov.pdf
- Matthay MA, Zemans RL, Zimmerman GA, Arabi YM, Beitler JR, Mercat A, et al. Acute respiratory distress syndrome. Nat Rev Dis Primer [Internet]. 2019 [cited 2020 May 14];5(1). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6709677/
- Here’s the Damage Coronavirus (COVID-19) Can Do to Your Lungs [Internet]. Health Essentials from Cleveland Clinic. 2020 [cited 2020 May 13]. Available from: https://health.clevelandclinic.org/heres-the-damage-coronavirus-covid-19-can-do-to-your-lungs/
- Sun P, Qie S, Liu Z, Ren J, Li K, Xi J. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: A single arm meta-analysis. J Med Virol. 2020 Feb 28;
- Gibson PG, Qin L, Puah S. COVID-19 ARDS: clinical features and differences to “usual” pre-COVID ARDS. Med J Aust. 2020 Apr 24;1.
- Zhang JJY, Lee KS, Ang LW, Leo YS, Young BE. Risk Factors of Severe Disease and Efficacy of Treatment in Patients Infected with COVID-19: A Systematic Review, Meta-Analysis and Meta-Regression Analysis. Clin Infect Dis Off Publ Infect Dis Soc Am. 2020 May 14;
- Santoso A, Pranata R, Wibowo A, Al-Farabi MJ, Huang I, Antariksa B. Cardiac injury is associated with mortality and critically ill pneumonia in COVID-19: A meta-analysis. Am J Emerg Med. 2020 Apr 19;
- OpenSAFELY: factors associated with COVID-19-related hospital death in the linked electronic health records of 17 million adult NHS patients. | medRxiv [Internet]. [cited 2020 May 24]. Available from: https://www.medrxiv.org/content/10.1101/2020.05.06.20092999v1
- National Academies of Sciences E, Division H and M, Practice B on PH and PH, States C on C-BS to PHE in the U, Baciu A, Negussie Y, et al. The State of Health Disparities in the United States [Internet]. Communities in Action: Pathways to Health Equity. National Academies Press (US); 2017 [cited 2020 Jun 1]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK425844/
- Huang I, Lim MA, Pranata R. Diabetes mellitus is associated with increased mortality and severity of disease in COVID-19 pneumonia – A systematic review, meta-analysis, and meta-regression. Diabetes Metab Syndr. 2020 Apr 17;14(4):395–403.
- Tamayo T, Brinks R, Hoyer A, Kuß O, Rathmann W. The Prevalence and Incidence of Diabetes in Germany. Dtsch Ärztebl Int. 2016 Mar;113(11):177–82.
- Seif F, Aazami H, Khoshmirsafa M, Kamali M, Mohsenzadegan M, Pornour M, et al. JAK Inhibition as a New Treatment Strategy for Patients with COVID-19. Int Arch Allergy Immunol. 2020 May 11;1–9.
- Azkur AK, Akdis M, Azkur D, Sokolowska M, van de Veen W, Brüggen M-C, et al. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in. Allergy. 2020 May 12;
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet. 2020;395(10229):1033–4.
- Bhargava P, Panda P, Ostwal V, Ramaswamy A. Repurposing valproate to prevent acute respiratory distress syndrome/acute lung injury in COVID-19: A review of immunomodulatory action. Cancer Res Stat Treat. 2020 Jan 4;3(5):65.
- Berger JR, Brandstadter R, Bar-Or A. COVID-19 and MS disease-modifying therapies. Neurol Neuroimmunol Neuroinflammation. 2020 Jul;7(4).

