Drug vignettes: Colchicine
February 3, 2021
Updated 19 January 2022
Robin E Ferner*†, Reecha Sofat†, Jeffrey K Aronson
Centre for Evidence-Based Medicine
Nuffield Department of Primary Care Health Sciences, University of Oxford
Correspondence to r.e.ferner@bham.ac.uk
*University of Birmingham †University College London
 Colchicine is an alkaloid present in all parts of the autumn crocus, Colchicum autumnale. It has been used therapeutically since the first half of the 19th century.
Colchicine is an alkaloid present in all parts of the autumn crocus, Colchicum autumnale. It has been used therapeutically since the first half of the 19th century.
Indications
In the UK colchicine is licensed for
- treatment of acute gout;
- prevention of acute gout during the early phases of treatment of hyperuricaemia (for example with allopurinol or uricosuric drugs).
Colchicine has also been used in the treatment of acute pericarditis, familial Mediterranean fever, and Behçet’s disease.
Mode of action
The mode of action of colchicine in gout is not known. A plausible explanation is that it inhibits migration of phagocytes to gouty tissues and thereby prevents both phagocytosis of urate crystals and the subsequent increase in local lactate release following phagocytosis, which lowers the pH and leads to further urate deposition. However, colchicine has other anti-inflammatory actions, including inhibition of cellular microtubular function and inhibition of the NLRP3 inflammosome complex. This can result in suppression of secretion of interleukins, including IL-1b, IL-6, and IL-18.
Dosages
- Acute gout: 1 mg orally initially, then 0.5 mg 1 hour later; colchicine should then not be given for a further 12 hours, after which it can be given in a dose of 0.5 mg every 8 hours until symptoms are relieved or until 6 mg has been given. After that the drug should not be given again for at least 3 days.
- Prophylaxis of acute gout: 0.5 mg twice daily by mouth.
Dosages should be reduced in hepatic failure and renal failure and patients should be carefully monitored for adverse effects. Avoid in severe hepatic or renal failure.
Colchicine should not be used in pregnancy or during breastfeeding.
Kinetic data
Colchicine is rapidly and well absorbed after oral administration. Although it is said to have a short half-life (3–10 h), in leukocytes it may be detectable for several days after a single dose, and clearly tissue accumulation occurs.
Colchicine is mostly metabolized (80%) in the liver and its metabolites are excreted in the bile.
Important adverse effects
- Gastrointestinal symptoms are dose-related within the therapeutic range of doses, and include abdominal pain, diarrhoea, nausea, and vomiting, and in severe poisoning an enteritis resembling cholera.
- Rarely bone-marrow suppression, resulting in leukopenia and thrombocytopenia, can occur during long-term administration.
- Steatorrhoea and peripheral neuropathy or myopathy can occur during long-term administration.
Drug–drug interactions
Colchicine is a substrate for both CYP3A4 and P glycoprotein. It should not be used in patients taking drugs that inhibit those systems, such as macrolide antibiotics, ciclosporin, antifungal imidazoles, HIV protease inhibitors, some calcium channel blockers (verapamil and diltiazem), and disulfiram.
Clinical studies
Retrospective studies
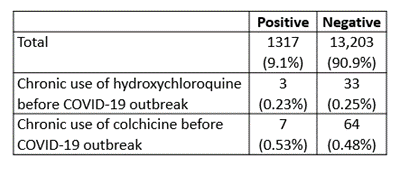
In a small retrospective database study from Israel early in the pandemic, the characteristics of 1317 patients positive for SARS-CoV-2 infection were compared with those in 13,203 negative patients. The rates of positivity were no different from average in those patients who took either hydroxychloroquine or colchicine long-term (Table 1). Hydroxychloroquine is ineffective in COVID-19.
Table 1. Results of Gendelman et al.
Randomized studies in hospitalized patients with COVID-19
GRECCO-19
GRECCO-19 was a small prospective, open, randomized clinical trial comparing the effects of standard care with the effects of colchicine (1.5 mg once, 0.5 mg at 1 hour, then 0.5 mg twice daily for up to 3 weeks, i.e. a total of 23 mg) in patients admitted to hospital with symptoms and signs of COVID-19 and PCR positive tests for SARS-CoV-2 (Table 2). Those who had a QT interval of 450 milliseconds or more, liver failure, or colchicine sensitivity, or were pregnant or breast-feeding were excluded, as were those deemed to need “ventilatory support”.
Table 2. Data at entry to GRECCO-19
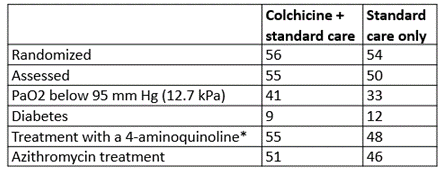 *Chloroquine or hydroxychloroquine
*Chloroquine or hydroxychloroquine
The co-primary end-points in the biochemical phase were the difference in maximal concentrations of high-sensitivity cardiac troponin (hs cTn) between the two groups and the time for C-reactive protein to reach values more than three times the upper limit of the reference range (not subsequently analysed).
The primary end-point of the clinical phase was the time from baseline to clinical deterioration, defined as a 2-grade increase on an ordinal clinical scale.
The results are shown in Table 3.
The authors concluded that “participants who received colchicine had statistically significant improved time to clinical deterioration [sic] compared with a control group that did not receive colchicine. However, the observed difference was based on a narrow margin of clinical significance; these observations should therefore be considered hypothesis generating.”
Table 3. Selected results from GRECCO-19; primary end-points shown in bold
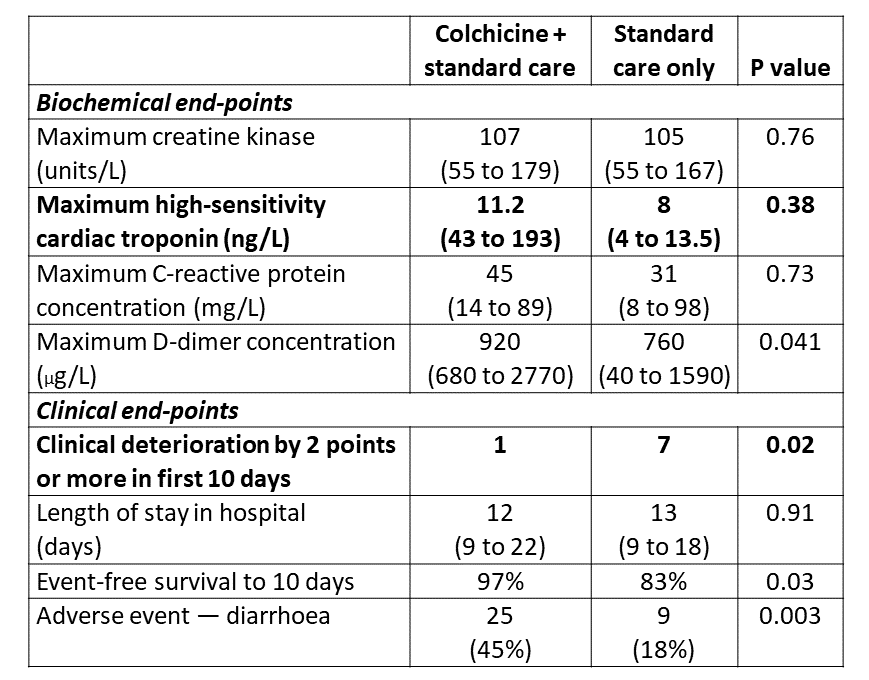
This was a small open study with some baseline differences between patients in the two arms. The number of patients recruited was nearly 40% less than the 180 patients stipulated in the (amended) protocol.
There was significant benefit from treatment when judged by changes on an ordinal scale, but ordinal scales are open to criticism, because a change at one part of the scale is not equivalent to the same change at another part of the scale.
The original hope was that colchicine would reduce cardiac damage from SARS-CoV-2, but this was not demonstrated.
The authors were suitably cautious in suggesting that the results are “hypothesis generating”.
Randomized studies in patients with COVID-19 who have not been admitted to hospital
COLCORONA
An international clinical trial, with a protocol that was published in March 2020, sought to establish whether colchicine given in early COVID-19 could reduce the chance of hospitalization or death. The COLCORONA trial set out to recruit 6000 patients with at least one risk factor, in whom COVID-19 had been diagnosed outside hospital within 24 hours of recruitment. Patients were randomized 1:1 to 30 days of treatment with colchicine (500 micrograms twice daily for three days, then once daily for 27 days, i.e. a total of 16.5 mg) or placebo. Recruitment began in March 2020.
A press release published on 23 January 2021 announced that the trial showed that “colchicine has reduced by 21% the risk of death or hospitalizations in patients with COVID-19 compared to placebo.” A preprint of the study has now been published.
Methods
The preprint describes the trial as “double-blind”, as did the first version of the protocol, published on 20 March 2020, and all subsequent versions. However, the same protocol (version 1) described masking as “Single (Participant)”. This was not changed, despite subsequent amendments, until 13 November 2020 (version 11), when masking was described as “Triple (Participant, Care Provider, Investigator)”.
Entry criteria:
Age 40 years or more
+ COVID-19 diagnosed within 24 hours of enrolment
+ Not currently hospitalized or considered for immediate hospitalization
+ One or more of the following risk factors:
o Age 70 years or more
o Body-mass index of 30 kg/m2 or more
o Diabetes [mellitus]
o Uncontrolled hypertension, systolic blood pressure at or above 150 mmHg
o Known respiratory disease
o Known heart failure or coronary disease
o Fever of at least 38.4oC within the last 48 hours
o Dyspnoea at presentation
o Defined haematological abnormalities (bicytopenia, pancytopenia, or
↑neutrophil count with ↓lymphocyte count)
The diagnosis of COVID-19 was made in one of three ways:
- Positive PCR on a nasopharyngeal swab
- Symptoms compatible with COVID-19 and a household member with a positive PCR test
- Symptoms without alternative causes and an algorithmic diagnosis of COVID-19
Patients were excluded if they were already taking colchicine or had inflammatory bowel disease, severe hepatic or renal impairment, or certain other conditions (set out in a Supplement that is not readily available).
Evaluation was made by telephone 15 and 30 days after recruitment.
The primary end-point was death or hospital admission within 30 days of randomization. Secondary end-points were death, hospital admission, or mechanical ventilation within the same time-frame. Intention-to-treat analyses were made by chi-squared tests.
A prespecified analysis of those with positive PCR tests was also made, and other subgroup analyses were conducted using logistic regression models.
While not mentioned in the protocol, the preprint states that a “pre-specified stopping rule for efficacy was based on the Lan-DeMets procedure with the O’Brien–Fleming alpha-spending function” — statistical procedures that allow for interim analysis. In fact, the decision to halt the study after recruitment and follow-up had reached 75% of the planned numbers was made “due to logistical issues related to maintaining the central study call center active 24 hours per day for a prolonged period of time, as well as the need to provide healthcare systems with study results in a timely fashion given the state of the COVID-19 pandemic.” The level of statistical significance was adjusted by 0.001 to 0.049 “to account for the interim analyses”. There was no adjustment for multiple comparisons.
Table 4. Data at entry into COLCORONA

Outcomes
The primary and planned secondary outcomes in the entire population and in those with positive PCR tests were as shown in Table 5.
Table 5. Selected results from COLCORONA

*The figures show that three patients in each group died but were not admitted to hospital
Adverse events
A diagnosis of pulmonary embolism was significantly more common in patients assigned to colchicine treatment than in those assigned to placebo [11 (0.5%) versus 2 (0.1%), P=0.01]. Patients assigned to colchicine treatment also suffered more diarrhoea (13.7% versus 7.3%, P<0.0001).
Comments
- The true extent of masking in the study is unclear, because the protocol up to November 2020 specified that only patients, not investigators or others, would be masked to the treatment, although the title described the trial as “double-blind”.
- The statistical techniques used suggest that interim analysis was pre-planned, but this was not signalled in the trial protocol.
- Recruitment was less than planned, for “logistic” reasons.
- The results of the planned primary analysis in all patients were not statistically significant.
- The prespecified number of participants requiring mechanical ventilation was not reported.
- Analysis of only those patients whose PCR test was positive for SARS-CoV-2 was not the primary hypothesis to be tested, and there was no adjustment of tests of significance for multiple testing.
- There was no justification for stopping the trial prematurely “to provide healthcare systems with study results in a timely fashion given the state of the COVID-19 pandemic”, since the results were inconclusive. What healthcare systems have been given is uncertainty.
Colchicine in hospital in-patients
The effects of colchicine in hospitalized patients with moderate or severe COVID-19, confirmed by RT-PCR and CT lung changes but not already requiring ICU admission, have been studied in a small randomized, double-blind, placebo-controlled Brazilian study. A preliminary report of the results in 38 patients was posted in medRxiv on 11 August 2020 and a second version a day later. Some of the details in these versions differed from those in the final report.
Patients who weighed under 80 kg were given colchicine in doses of 500 micrograms three times daily for 5 days, then 500 micrograms twice daily for a further 5 days (i.e. a total of 12.5 mg). In those who weighed 80 kg or more, a loading dose of 1 mg was given. All received standard care with azithromycin, hydroxychloroquine, and unfractionated heparin; part way through the study methylprednisolone was added.
The primary endpoints were rather diffusely defined as “clinical parameters, such as the time of need for supplemental oxygen; time of hospitalisation; need for admission and length of stay in ICU; and death rate and causes of mortality.” Laboratory results were was also assessed, as were adverse events, including the frequency of QT interval prolongation beyond 450 ms.
Selected results from the trial are shown in Table 6.
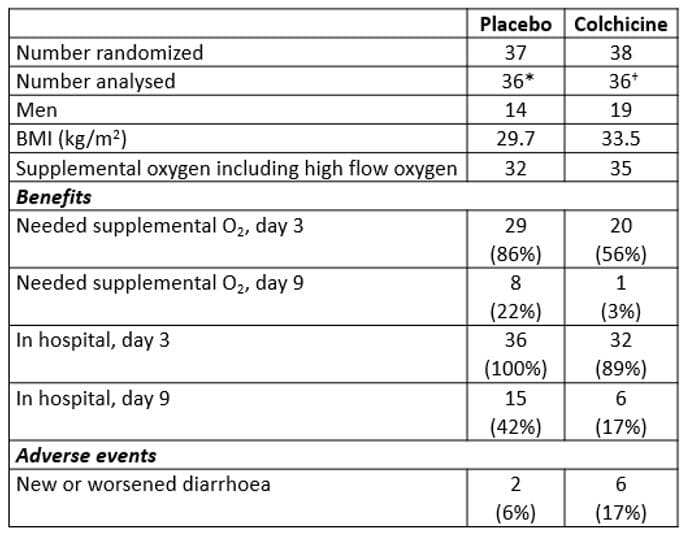
*One admitted to ICU
†One ventilated before treatment, one admitted to ICU
The authors stated that there were significant benefits, judged by the need for supplemental oxygen (log rank test 10.6, P=0.001) and for length of hospital stay (log rank test, 9.2, P=0.002).
Comments
This was a very small trial.
The primary endpoints were poorly specified.
Deaths and ICU admissions yielded too few events for statistical analysis.
The trial departed from one published protocol, but more closely followed another.
Diarrhoea again emerges as an adverse event associated with colchicine.
The authors concluded, reasonably, and as authors of small trials so often do, that “Clinical trials with larger numbers of patients should be conducted to further evaluate the efficacy and safety of colchicine as an adjunctive therapy for hospitalized patients with moderate to severe COVID-19.”
Registered trials of colchicine in COVID-19
We currently know of 36 registered trials involving colchicine, alone or in combination with other treatments, of which 35 are randomized and 21 are recorded to be recruiting. Twenty-five are not masked (mostly described as “open label”); two are single-blind (participants only), five double-blind, two triple-blind, and one quadruple-blind. Of the trials that are recruiting and have declared the number of subjects they aim to enrol, four specify under 100 subjects, six between 100 and 1000, and two over 1000. Taking all the trials, the total number of participants proposed to be enrolled in double-, triple-, or quadruple-blind trials is 6994 (22%) out of a total of 32,097.
Outlook
The efficacy of colchicine in patients with mild or moderate COVID-19 remains uncertain after these inconclusive studies. Adverse effects from treatment with colchicine for a month are significant, and the total doses that have been used are very high. Until better evidence emerges, colchicine is better reserved for use in randomized studies.
Update added on 19 January 2022
Three further trials have provided evidence that colchicine is unlikely to be effective in treating patients with COVID-19 in the community or in hospital.
RECOVERY
The RECOVERY investigators randomly assigned 11 340 patients with clinically suspected or laboratory confirmed SARS-CoV-2 infection to usual care alone or usual care plus colchicine in a large multicentre trial. The primary endpoint was death within 28 days of randomization.
The dose of colchicine was 1 mg followed by 0.5 mg after 12 hours and then 0.5 mg twice daily for 10 days. Recruitment ended in March 2021.
The groups were well balanced for age, sex, ethnicity, duration of symptoms, and concomitant disorders. In both groups 97% of the patients had positive PCR tests for SARS-CoV-2. Over 90% in both groups received dexamethasone.
“There was no significant difference in the proportion of patients who died within 28-days between the two groups (1173 [21%] in the colchicine group vs 1190 [21%] in the usual care group; rate ratio 1·01 [95% CI 0·93–1·10]; P=0·77).” Nor were there statistically significant differences in outcome for prespecified subgroups or secondary outcomes.
Limitations: A further 8083 patients were excluded, either because colchicine was unavailable (1072), or because the patients were considered unsuitable (7688). Unsuitable patients included children, pregnant women, those with severe hepatic impairment, significant cytopenias, or lactose intolerance, and those taking drugs that might interact.
The trial was open-label; the investigators assessed outcomes from routine healthcare records.
The trial was stopped after an independent data monitoring committee found that “there was no convincing evidence that continued recruitment to the colchicine comparison would provide conclusive proof of worthwhile mortality benefit either overall or in any prespecified subgroup.”
COLCOVID
The COLCOVID trial was a multicentre, open-label, randomized clinical trial in hospital patients with confirmed or suspected SARS-CoV-2 infection followed for up to 28 days. The investigators randomized 640 patients to receive colchicine and 639 patients to receive usual care. The primary endpoint was death by 28 days or mechanical ventilation.
Colchicine was given in a loading dose of 1.5 mg immediately after randomization, followed by 0.5 mg orally within 2 hours of the initial dose, and then 0.5 mg orally twice a day for 14 days or until discharge.
The primary endpoint was reached by 160 (25%) of the colchicine group and 184 (29%) of the usual care group, hazard ratio 0.83, 95% CI 0.67–1.02, P=0.08; 131 patients (21%) assigned to colchicine and 142 patients (22%) assigned to usual care died within 28 days of randomization, hazard ratio 0.88, 95% CI 0.70–1.12.
Severe diarrhoea was reported by 11% of those who received colchicine and 4.5% of those who received placebo.
Limitations: The protocol initially aimed to recruit 2500 patients, and defined the primary outcome as in-hospital mortality, but was amended because recruitment was slow. The trial was designed to have statistical power to detect a difference of 27% between the groups. The observed difference was 17%, which was not statistically significant, but which might imply a difference in the primary outcome. The trial was open-label.
COLCHIVID
In the COLCHIVID trial, a triple-blind parallel non-stratified placebo-controlled trial, 116 hospitalized patients with severe COVID-19 were randomized to 1.5 mg of colchicine (n=56) or placebo (n=60) at the time of recruitment followed by 0.5 mg twice daily for 10 days. The primary composite outcome reported was the progression to critical disease or death.
The study was stopped after the second interim analysis showed that colchicine had no effect on the reported primary outcome (OR = 0.83, 95%CI = 0.35-1.93, P = 0.67), or on the number of days spent in ICU or in the hospital overall. Nor did it have any effect on other prespecified primary outcomes, such as fever, myalgia, or arthralgia. Adverse events were similar between the groups (OR = 1.63, 95% CI = 0.66-3.88, P = 0.37); eight patients who received colchicine had diarrhoea compared with four who received placebo.
Limitations: This was a very small study, with 96% power to detect a 50% reduction in mortality, a very large reduction. The protocol specified 174 participants, but the trial was stopped early. The authors’ conclusion that colchicine was “safe” was not fully justified, especially in the light of outcomes in other larger studies.
Meta-analysis
In addition to the more recent randomized trials, a meta-analysis of six studies with a total of 16 148 patients with suspected or proven COVID-19, showed no statistically significant benefit from colchicine. The RECOVERY and COLCORONA trials contributed 98% of the patients, the other four trials (1,2,3,4) being very small. The risk difference for mortality was 0.0 (95% CI –0.01 to 0.01). The relative risk of diarrhoea was 1.93 (95% CI 1.62 to 2.29).
Outlook on 30 December 2021: Our previous conclusion was that colchicine is better reserved for use in randomized trials. The currently available results from such trials provide no convincing evidence that colchicine will save lives, although the studies so far cannot rule out a small but clinically significant effect on mortality. If colchicine were used in further clinical trials in potentially effective doses, more than 10% of patients would develop severe diarrhoea.

