Drug vignettes: Interferons
August 6, 2020

Jeffrey K Aronson, Nicholas DeVito, Annette Plüddemann, Robin E Ferner*†
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
*University of Birmingham
†University College London
Correspondence to jeffrey.aronson@phc.ox.ac.uk
Description of the drug
Interferons (IFNs) were first described in 1957 by Isaacs and Lindenmann, who observed secretion of a novel substance when live influenza virus in chick chorio-allantoic membranes was exposed to heat-inactivated virus [1]. They called the substance “interferon”.
Interferons are cytokines with well described antiviral properties, released from host cells in response to the aberrant nucleic acids that characterize viral infections, acting through host toll-like receptors (TLRs) and other pattern-recognition receptors that are responsive to viral RNA or DNA [2]. With other cytokines, interferons are responsible for some of the symptoms of viral infection, including fever and muscle pain. Microbial products and chemical inducers can also stimulate interferon production [3]. Interferons also have effects on tumour cell survival via TLRs [4].
There are three types of interferons, I, II, and III. Interferons I (IFN-α and IFN-β) and interferon II (IFN-γ) are used therapeutically (Table 1).

Type I interferons
There are several subtypes of type I interferons, identified by Greek suffixes, including IFN-α, IFN-β, IFN-κ, and IFN-ω [5]. Only the first two of these are used therapeutically.
Type I interferons promote the function of natural killer cells, B cells, T cells, and myeloid dendritic cells [6] and upregulate the expression of MHC antigens [7].
Type I interferons are produced by a range of cells, including macrophages, plasmacytoid dendritic cells, monocytes, fibroblasts, and microglial cells [8]. Subtypes of these interferons differ from each other minimally; for example, IFN-α2a and IFN-α2b differ by a single amino acid at position 23 in a 166-amino acid sequence.
INF-β is a glycosylated protein of 166 amino acids. IFN-β1a for therapeutic use is produced in Chinese hamster ovary cells by recombinant DNA technology. In the UK there are licensed subcutaneous formulations, but no licensed formulations for inhalation.
Interferon type II
Interferon II is known as IFN-γ or immune interferon and is activated by interleukin 12 (IL-12). It is also released by cytotoxic T cells and type 1 T-helper cells (Th1). It blocks the proliferation of type 2 T-helper cells (Th2).
Mechanism of action
Production of interferons requires activation of intracellular interferon regulatory factors (IRFs), of which nine human types have been described [9]. When pattern recognition receptors recognize pathogen-associated molecular patterns in the cytosol, they stimulate adaptor proteins that activate serine/threonine kinases that phosphorylate IRFs3 and IRF7; this allow them to be translocated into the nucleus. There, IRFs, in combination with the transcription factor NF-κB, induce transcription of IFN-β1 and increase its production [10].
IFN-α and IFN-β bind to IFN-α/β transmembrane receptors (IFNARs), activating intracellular signalling through the Janus kinase signal transducer and activator of transcription (JAK-STAT) pathway and other pathways [11]. This results in expression of very large numbers of genes required for host defences against infections [12]. IFN-γ binds to the IFN-γ receptor (IFNGR or IFN-γR), which also acts via JAK-STAT.
The main signalling pathways for IFN-I (IFN-α and IFN-β), and IFN II (IFN-γ) are shown in Figure 1.
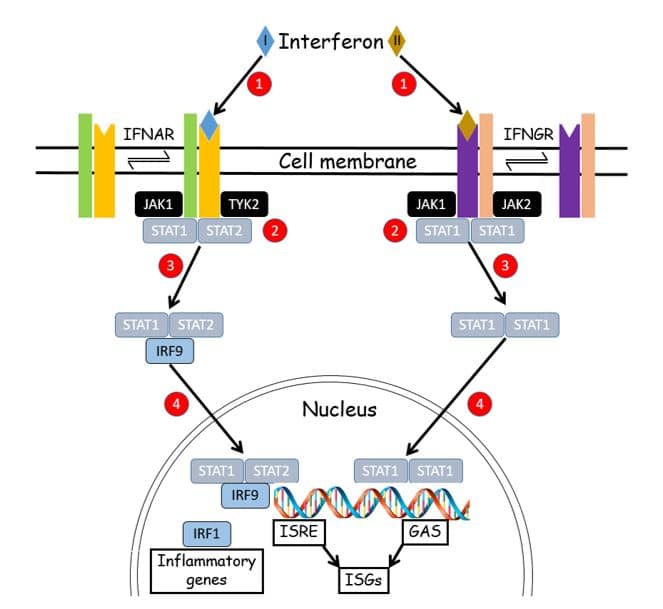
Figure 1. The main signalling pathways for Type I interferons (IFN-α and IFN-β) and Type II interferon (IFN-γ).
- Interferons, released from secretory cells, bind to receptors in the cell membrane
- They activate JAK-STAT pathways
- After phosphorylation by Janus and tyrosine kinases, STAT molecules bind to interferon regulatory factors (IRFs) and enter the nucleus; in the case of IFN-I they switch on Interferon-Stimulated Genes (ISGs), via Interferon-Stimulated Response Elements (ISRE), and inflammatory genes, which express cytokines and chemokines; in the case of IFN-II they stimulate gamma-activated sequences (GAS) in the promoter regions of ISGs
Practical use in licensed indications
Most available formulations are for subcutaneous injection. One commercial formulation of recombinant IFN-β1a (Avonex®) and three of recombinant IFN-β1b (Betaferon®, Extavia®, and Rebif®) are licensed in the UK. They are variously indicated for the treatment of patients with different types of multiple sclerosis (Table 2). It is probable that the clinical benefit is small [13], and there are more effective treatments [14].
A formulation of rIFN-γ1b (Immukin®) is licensed in the UK for the treatment of severe infection in patients with chronic granulomatous disease.

A patent was granted in 2005 to the University of Southampton and others for “Anti-virus therapy for Respiratory Diseases” by airway delivery of IFN-β or an agent that increases IFN-β expression [16,17]. A formulation of rIFN-β1a for inhalation (code named SNG001, manufactured by Synairgen Research Limited, a spin-off company of the University of Southampton) has been in development since at least 2010. It has been trialled in prevention or attenuation of symptoms of chronic obstructive pulmonary disease (COPD) caused by respiratory viruses, according to a protocol listed in the EU Clinical Trials Register [18]. However, it is not yet licensed.*International units of interferon activity are measured by comparing test preparations against a standard in assays that measure inhibition of viral cytopathic effect or quantify viral reporter proteins [15]; correspondence between milligrams and MIU (million international units of activity) differs from type to type; for example (data from Summaries of Product Characteristics):
IFN-β1a: 1 mg = 273 MIU
Pegylated IFN-β1a: 1 mg = 95 MIU
Recombinant IFN-β1b: 1 mg = 32 MIU
Recombinant human IFN-γ1b: 1 mg = 20 MIU
More recently the same company has trialled SNG001 in patients with COVID-19 [19].
Pharmacokinetics
Intravenously administered interferons pass directly from the bloodstream into the tissues. In contrast, after subcutaneous administration, the commonly used route, they enter the lymphatics and are slowly absorbed [20].
The plasma half-life after intravenous administration is about 8 hours. After subcutaneous administration of 18 MIU, the maximum plasma concentration is 5 IU/L; slow absorption limits the elimination rate, and the observed half-life is about 17 hours [21]. From intravenous data the calculated clearance is about 350 mL/min and the apparent volume of distribution about 250 L.
Only about a third of a dose of interferon is absorbed after subcutaneous administration. Pegylated formulations are absorbed more completely (about 85%) [22]; the rate-limited terminal half-life after subcutaneous administration of pegylated IFN-α2b was 43–51 hours.
When interferons are given intramuscularly, antiviral activity is maximal at 5–10 hours after administration [23] and the terminal half-life is 50–60 hours [24].
However, the pharmacokinetics of inhaled IFN-β1a are unclear. No circulating IFN-β activity was found after inhalation of up to 100 MIU by eight patients with lung cancer [25], which suggests that after inhalation it is absorbed into the systemic circulation poorly or not at all.
Known contraindications and cautions
The licensed subcutaneous formulations are contraindicated in patients with current severe depression or suicidal ideation or both.
Known and potential adverse effects/adverse reactions
In 21 healthy volunteers who inhaled a formulation of IFN-β1a 300 micrograms once a week for four weeks there was a significant reduction of about 12% in lung carbon monoxide transfer factor [26].
The many listed adverse effects of subcutaneously administered IFN-β include:
Very common Asymptomatic increases in transaminases; headache; injection site reactions, influenza-like symptoms; neutropenia, lymphopenia, leukopenia, thrombocytopenia, anaemia.
Common Depression, insomnia; diarrhoea, vomiting, nausea; injection site pain, fatigue, rigors, fever; myalgia, arthralgia; pruritus, erythematous and maculopapular rashes, alopecia; large rises in transaminases.
Uncommon Dyspnoea; hepatitis; injection site necrosis, abscesses, and infections; increased sweating; retinal vascular disorders (retinopathy, cotton wool spots, retinal artery or vein obstruction); seizures; thromboembolic events; thyroid dysfunction; urticaria.
Rare Anaphylactic reactions; lupus-like reactions; hepatic failure, autoimmune hepatitis; injection site cellulitis; nephrotic syndrome, glomerulosclerosis; angioedema, erythema multiforme, erythema multiforme-like skin reactions, Stevens–Johnson syndrome; suicide attempt; thrombotic microangiopathy including thrombotic thrombocytopenic purpura/haemolytic–uraemic syndrome, pancytopenia.
Frequency not known Panniculitis at the injection site); pulmonary arterial hypertension; transient neurological symptoms (hypaesthesia, muscle spasm, paraesthesia, difficulty in walking, musculoskeletal stiffness), which may mimic exacerbations of multiple sclerosis.
It is not known whether the adverse effects of subcutaneous IFN-β are relevant to those of IFN-β administered by inhalation.
Drug-drug interactions
Interferons inhibit the hepatic microsomal cytochrome P450 enzyme CYP1A2 in vitro and in vivo [27].
Alcohol
Exacerbation of previous acute alcohol hepatitis has been reported in two patients with chronic hepatitis C, despite reduced alcohol consumption; hepatic aminotransferases normalized after withdrawal of IFN-α in both patients [28]. Alcohol may reduce the efficacy of interferon in hepatitis C [29].
Angiotensin-converting enzyme inhibitors
An increased risk of severe and early but reversible neutropenia has been reported in patients taking enalapril and captopril with IFN-α [30].
Carmustine
In 275 patients randomized to receive radiation and carmustine, either alone or with IFN-α, for high-grade glioma, there was no significant improvement in the overall survival and time to disease progression in those given IFN-α, but a higher incidence of adverse reactions, namely fever, chills, myalgia, lethargy, headache, and seizures [31].
Coumarin anticoagulants
There have been two reports of increased prothrombin time when patients taking warfarin or acenocoumarol were also given IFN-α [32,33].
Cyclophosphamide
Depending on the timing of exposure, IFN-α may adversely affect the pharmacokinetic and haematological effects of cyclophosphamide. In 10 patients with multiple myeloma, IFN-α given 2 hours before cyclophosphamide infusion significantly reduced cyclophosphamide clearance and produced less exposure to its metabolite 4-hydroxycyclophosphamide compared with interferon administration 24 hours after cyclophosphamide [34]. This resulted in a significantly greater fall in white blood cell count in patients who received IFN-α after cyclophosphamide.
Erythropoietin
Reduced efficacy of human erythropoietin, requiring increased erythropoietin dosages, has been reported in several patients receiving IFN-α [35,36,37], an effect that is probably mediated by IFN-α-induced suppression of erythropoiesis.
Fluorouracil
The combination of IFN-α with 5-fluorouracil produced increased serum concentrations of fluorouracil and a significantly higher incidence of severe adverse reactions due to gastrointestinal and myelosuppressive adverse effects [38,39]. IFN-α2b has been associated with an 80% increase in fluorouracil AUC, partly by reducing its clearance [40].
Natalizumab
Interferon treatment may increase the risk of progressive multifocal leukoencephalopathy in patients receiving natalizumab [41].
Ribavirin
Two patients developed rapid and particularly severe anaemia within 4 and 6 weeks of combined treatment with IFN-α and ribavirin [42]. One required erythrocyte transfusions and both recovered after withdrawal. The combination of pure red cell aplasia due to IFN-α and haemolytic anaemia due to ribavirin was suggested to have accounted for this possible interaction. There was an increased incidence of adverse skin reactions, mostly eczema, malar erythema, and lichenoid eruptions, in 33 patients who received combination of IFN-α with ribavirin compared with 35 patients treated with IFN-α alone [43].
Telbivudine
In a clinical trial of a combination of telbivudine 600 mg daily with subcutaneous pegylated IFN-α2a, 180 micrograms once weekly for chronic hepatitis B, there was an increased risk of peripheral neuropathy. The manufacturers state that the combination of Pegasys with telbivudine is contraindicated [44].
Thalidomide
In 13 patients with metastatic renal cell carcinoma, the combination of IFN-α2a (27 MIU/week) and thalidomide produced severe neurological toxicity in four patients, an incidence that was considered to be far greater than would be expected with either drug alone [45].
Theophylline
Both IFN-α [46,47,48,49] and IFN-β [50] reduce theophylline clearance.
Resistance
Many viruses have developed mechanisms of resistance to interferons or block their actions, including adenoviruses, hepatitis C, influenza and parainfluenza viruses, Japanese encephalitis virus, and paramyxoviruses [51,52].
Experience in coronavirus infections other than COVID-19
In vitro studies
IFN-β1a was a potent inhibitor of SARS-CoV-1 in cell culture when applied before or 1 hour after inoculation, reducing viral replication by 2 log units [53].
Animal studies
Wild-type mice develop a mild bronchiolitis when infected with SARS-CoV-1 (which causes SARS), but Stat1 –/– mice, who lack the STAT pathway, have increased lung damage, continued viral replication, and damage to other organs [54].
Clinical studies
In a retrospective survey of 51 patients with MERS-CoV infection, 23 of whom were treated with IFN-β, survival was more likely in the patients treated with IFN-β. It is likely that it was given subcutaneously. As treatments were not allocated randomly, the result is not helpful in determining the possible therapeutic benefit of IFN-β [55].
The authors of an observational study in 349 patients with MERS, of whom 141 received ribavirin or an interferon or both, concluded that “In this observational study, RBV/rIFN (RBV and/or rIFN-α2a, rIFN-α2b, or rIFN-β1a) therapy was commonly used in critically ill MERS patients, but was not associated with reduction in 90-day mortality or in faster MERS-CoV RNA clearance.” However, 95 patients received one or another form of IFN-α, and only 31 patients were treated with IFN-β [56]. The protocol for a subsequent study has been published, and the trial is said to have been completed, but no results have been posted [57].
Experience in COVID-19
Justification
The aim of using interferons to treat COVID-19 is to boost the immune response to the infection, at least in the early phases of the disease. However, there is a risk that boosting the immune response in severe infection might be counterproductive, and a better strategy would be to inhibit it, as evidenced by the efficacy of dexamethasone [58].
The usual host response to viral infection includes synthesis of interferon after the virus has entered cells and started to replicate [59]. Intracellular pattern recognition receptors detect abnormal RNA and activate cellular transcription factors, including interferon regulator factors (IRFs) and nuclear factor κB (NF-κB). This leads to induction of IFN-I and IFN-III, followed by upregulation of IFN-stimulated genes. Leukocyte recruitment follows. However, in COVID-19 there is an inappropriate inflammatory response, in which there are low concentrations of IFN-I and IFN-III; in contrast TNF-α and IL-6 responses are enhanced compared with other types of viral infection, independent of IFN signalling [60,61]. Consistent with this, lymphocytopenia is a hallmark of disease intensity.
There have also been reports of young men in whom COVID-19 was severe, who turned out to have genetic polymorphisms associated with impaired IFN responses [62].
These observations appear to justify trials of interferons in COVID-19.
In vitro studies
IFN-β1a inhibited the growth of SAR-CoV-2 in vitro in Vero E6 cells [63]. After 72 hours exposure to the virus, complete inhibition was obtainable by the subsequent addition of IFN-β1a 10 IU/mL, with an EC50 of 2 IU/mL; after 96 hours exposure to the virus a higher concentration of subsequently added IFN-β1a was needed (4.7 IU/mL). In a similar study the EC50 values of IFN-α and IFN-β in Vero cells were 1.35 IU/ml and 0.76 IU/ml respectively at 24–48 after infection [64].
Clinical studies
IFN-α2b
A Chinese cohort of 77 PCR-positive patients received treatment with inhaled nebulized IFN-α2b 5 MIU twice daily (7 patients) or oral umifenovir (Arbidol) (24 patients), or both (46 patients) [65]. None required prolonged oxygen therapy or ventilation. Compared with umifenovir alone, any inhaled IFN-α2b treatment significantly increased the rate of viral clearance by about 1 week. However, as the authors said, “This exploratory study has several significant limitations. Most obvious is the fact that the study cohort was small, nonrandomized, with unbalanced demographics between treatment arms that were of unequal size.” Corticosteroid use in this study was not mentioned.
In a prospective multicenter observational study in Cuba, 814 patients with confirmed SARS-CoV-2 infection received therapy with a combination of oral antivirals (lopinavir/ritonavir and chloroquine) and intramuscular IFN-α2b 3 times a week for 2 weeks [66]; 761 received IFN and 53 did not. The proportion of fully recovered patients was higher in the former (95% vs 26%, P<0.01). The overall case fatality rate was 2.95%, and 0.92% in those who received IFN-α2b. Of 82 patients who required intensive care, 42 received IFN-α2b. Corticosteroid use in this study was not mentioned. [Not peer reviewed.]
IFN-β1a
In an unmasked randomized clinical trial from Tehran, 42 patients received subcutaneous IFN-β1a 44 micrograms three times a week for two consecutive weeks, in addition to standard treatment (hydroxychloroquine plus lopinavir/ritonavir or atazanavir/ritonavir); 39 patients received standard treatment alone [67]. Corticosteroids and antibiotics were “recommended in the hospital protocol if indicated”. Only 64% of the patients were PCR-positive. The primary outcome was “time to reach clinical response”, as judged by an improvement of two points on the six point clinical severity scale, or discharge from hospital. As the primary outcome, time to the clinical response was not significantly different between the IFN-β1a and the control groups (9.7 ± 5.8 vs. 8.3 ± 4.9 days respectively, P=0.95). However, the 28-day mortality was reduced in the IFN-β1a group: 19% vs. 44% in controls, P=0.015.
In an Iranian study, 20 PCR-positive SARS-CoV-2 patients (of 64 assessed for eligibility) were given subcutaneous IFN-β1a 44 micrograms on alternate days for up to five doses [68]. They all also received hydroxychloroquine and lopinavir-ritonavir. Three patients received non-invasive ventilation for hypoxia. Ninety percent of patients were PCR-negative (Ct value at or above 37) at day 10 of treatment. Malaise persisted until day 14 in half the patients. The authors concluded that “the net therapeutic effect of IFN-β1a along with all other drugs may not be established exactly.” Corticosteroid use in this study was not mentioned.
IFN-β1b:
Five Korean patients with COVID-19 severe enough to require supplementary oxygen improved when they were given lopinavir/ritonavir, hydroxychloroquine, and IFN-β1b by an unstated route. Four of the patients were also given methylprednisolone. This study provided no information on the efficacy of IFN-β [69].
In a multicentre, prospective, unmasked, randomized trial in adults with COVID-19 in Hong Kong 127 patients were randomly assigned (2:1) to a 14-day combination of lopinavir 400 mg + ritonavir 100 mg every 12 h, ribavirin 400 mg every 12 h, and three doses of IFN-β1b, 8 MIU on alternate days or to 14 days of lopinavir 400 mg + ritonavir 100 mg every 12 h (control group) [70]. The primary endpoint was the time to providing a nasopharyngeal swab negative for SARS-CoV-2. The combination group had a significantly shorter median time from the start of treatment to negative nasopharyngeal swab (7 days [IQR 5–11]) than the control group (12 days [8–15]; hazard ratio = 4·37 [95% CI 1·86–10·24]; P=0·0010). Adverse events included self-limiting nausea and diarrhoea, with no difference between the two groups. One patient in the control group stopped taking lopinavir–ritonavir because of biochemical hepatitis. No patients died during the study. A few patients were also given hydrocortisone, which may have been a confounding factor. This study provided no information about the contribution of IFN-β1b alone to the outcome.
The effect of IFN-β1b on in-hospital mortality has been studied retrospectively in a Spanish cohort study of 256 patients with moderate to severe pneumonia [71]. Some patients received corticosteroids (26%) and other treatments, such as hydroxychloroquine, lopinavir/ritonavir, and azithromycin. Overall mortality was 25% (63/256). Twenty-two of 106 patients (21%) in the interferon group died and 41 of 150 (27%) in the control group. This difference was not significant. The groups were not equally balanced; those who did not receive IFN-β1b had more comorbidities. [Non-peer-reviewed.]
Inhaled IFN-β1a
A company-sponsored study, whose results were reported in a press release on 20 July 2020, examined the effects of SNG001, a formulation of IFN-β1a for inhalation administered by nebulizer, in a randomized, masked, placebo-controlled, trial in 101 patients with COVID-19 who had been symptomatic for an average of 9.7 days [72]. This Phase II study was begun on 16 March but was not registered with clinicaltrials.gov until 12 May 2020, with an estimated intended enrolment of 400 patients [73]. The title given in the protocol was “A Randomised Double-blind Placebo-controlled Trial to Determine the Safety and Efficacy of Inhaled SNG001 (IFN-β1a for Nebulisation) for the Treatment of Patients With Confirmed SARS-CoV-2 Infection”. The results reported in the press release are shown in Table 3.
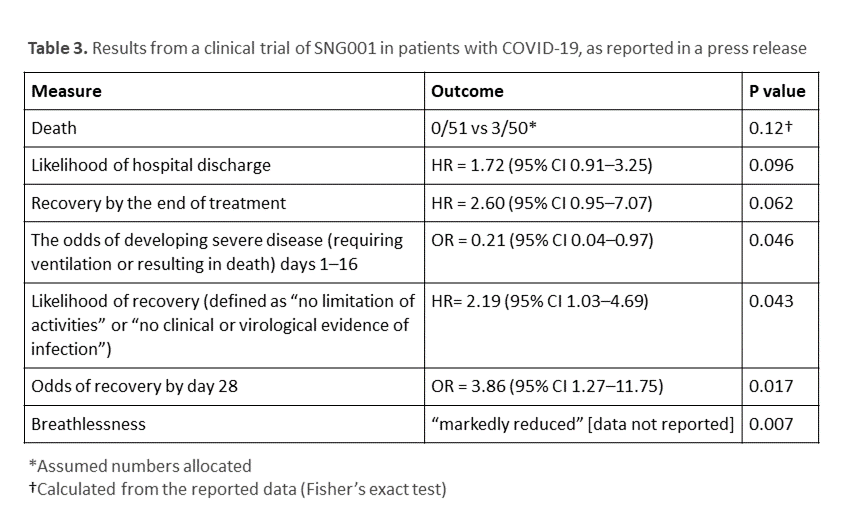
There are problems with these selectively reported results:
- Only 101 patients were randomized. This means that either the trial was terminated prematurely or interim results have been reported. A data monitoring committee could have mandated the former, but no such committee was mentioned in the protocol.
- The primary end-point specified in the protocol was “Change in condition measured using the Ordinal Scale for Clinical Improvement during the dosing period – minimum of 0 (patient is well) to a maximum of 8” from day 1 to day 28. However, the press release reported that “patients receiving SNG001 appeared to be more than twice as likely to have recovered by the end of the treatment period (HR 2.60 [95% CI 0.95–7.07]; p=0.062), although this strong trend did not reach statistical significance.” The patients were treated for 16 days.
- The press release stated that the treatment significantly reduced the odds of developing disease severe enough to be fatal or require ventilation, with an odds ratio of 0.21. The 95% confidence interval for this odds ratio was 0.04–0.97, i.e. from 1/5 to 5 times the mean value and the P value was only just below 0.05.
- Of 12 secondary endpoints, including seven describing “safety and tolerability”, only the beneficial effect on breathlessness was reported.
- No Bonferroni or other correction for multiple comparisons was reported in the brief press release, which quoted 95% confidence intervals.
- Use of corticosteroids, known to be beneficial, was not mentioned.
The scant trial data selected for the press release do not provide convincing evidence of the efficacy of inhaled IFN-β1a. In any case, much larger trials would be needed before we could be confident about the efficacy of nebulized IFN-β1a in COVID-19.
Registered clinical trials
We have identified 50 trials involving the use of interferons in the treatment of COVID-19, currently listed in a variety of registries; 23 are listed in ClinicalTrials.gov, 11 in the Iranian Registry of Clinical Trials, 11 in the Chinese Clinical Trials Registry, two in the Cuban Public Registry of Clinical Trials (RPCEC), and one each in the EU Clinical Trials Register, the Clinical Trials Registry—India, and the ISRCTN Registry.
Seven are triple- or quadruple-masked and two are single-masked (investigator or outcomes assessor). Most of them are small; 30 of the trials plan to recruit no more than 100 patients. Of 21,502 patients researchers intend to enrol, only 786 (3.7%) are in properly masked studies.
Most use standard care as a comparator and only nine involve a placebo; 15 that do not include standard care involve active comparators, mostly antiviral drugs, such as danoprevir, darunavir, favipiravir, lopinavir/ritonavir, ribavirin, and umifenovir.
Most of the published protocols specify the interferon to be used and all are of Type I. The subcutaneous route is specified in 20 cases and inhalational routes in 17; one involves eyedrops.
Conclusions
Despite the mechanistic rationale, interferons have not, up to now, proved useful treatments for acute virus infections. So far the data from small studies, only one of which appears to have been masked and randomized satisfactorily, show few beneficial effects and no convincing effect on mortality. Large, masked, randomized controlled trials would be needed to show whether inhaled or any other form of interferon reduces mortality in COVID-19. Only three trials currently planned are likely to be large enough and none of them is masked.
Disclaimer: This article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.

