Mast cell stabilisers, leukotriene antagonists and antihistamines: A rapid review of the evidence for their use in COVID-19
May 18, 2020

Meriel Raymond, Gemma Ching-A-Sue, Oliver Van Hecke
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
Correspondence to meriel.raymond@phc.ox.ac.uk
VERDICT
- Mast cells are the main source of cytokine release that leads to lung damage in SARS-CoV-2. It has been speculated that mast cell stabilisers may also attenuate pulmonary complications, fatal inflammation and death in COVID-19.
- At the time of writing, there have been no studies examining the use of mast cell stabilisers, leukotriene antagonists or anti-histamines in COVID-19.
- Clinical trials would be required to establish whether these drugs may be repurposed for treatment of this disease, though it may be preferable to channel resources towards therapeutics with a stronger evidence base.
BACKGROUND
Mast cells and the inflammatory response
Mast cells are present in mucosal and connective tissues throughout the body. They participate in the innate and adaptive immune process and have an important role in inflammation and allergy. Mast cells located in the submucosa of the respiratory tract are known to be activated by the SARS-CoV-2 virus (1) (see Figure 1). Once activated, degranulation occurs releasing histamine and proteases. Late mast-cell activation leads to the release of pro-inflammatory cytokines such as IL-1, IL-6 (1,2) and TNF-α (3).
Vitamin D is required to maintain the stability of mast cells, and Vitamin D deficiency results in mast cell activation(4).
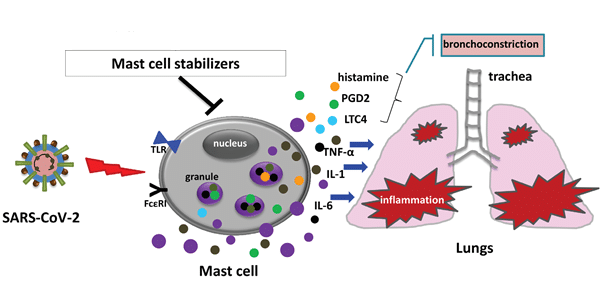
Figure 1 (5) (reproduced with author’s permission) SARS-CoV-2 may activate mast cells in the respiratory system through toll-like receptors (TLR) or by inducing the cross-linking of the IgE-FcεRI. Activated mast cells release pro-inflammatory cytokines such as IL-1, IL-6 and TNF-α as well as bronchoconstrictor mediators such as histamine, prostaglandin-D2 and leukotriene-C4. Abnormal production and release of these mediators from mast cells can further exacerbate SARS-CoV-2-induced inflammation in the respiratory system. Stabilisers of mast cells may alleviate inflammatory responses and pulmonary complications by suppressing activation of mast cells in SARS-CoV-2 infection. TNF-α: tumour necrosis factor-α, IL: interleukin, LTC4: leukotriene-C4, PGD2: prostaglandin-D2.
Current uses of mast cell stabilisers, leukotriene antagonists and antihistamines
All three classes of drugs are commonly used in the prevention and treatment of mast cell activation-mediated disorders ranging from asthma to allergic rhinitis. Leukotriene antagonists such as montelukast are widely used in the management of chronic asthma (6). Anti-histamines block airway inflammation and bronchoconstriction provoked by histamine release from mast cells, and are widely used in allergic conjunctivitis, rhinitis and sinusitis. Mast cell stabilisers e.g. sodium cromoglicate and ketotifen, inhibit degranulation of mast cells, and are used in asthma, allergic rhinitis and food allergy.
WHY CONSIDER THESE DRUGS IN COVID-19?
Symptom overlap
COVID-19 primarily presents in the respiratory system (7). Predominant symptoms are cough and fever; later complications include dyspnoea, pneumonia, respiratory failure and sepsis (a state where an infection from a micro-organism spreads throughout the body, causing an overwhelming inflammatory response, multi-organ damage and shock), which can be fatal (7). Allergic conditions of the respiratory tract such as rhinitis and asthma can cause rhinorrhoea, cough and breathlessness which are symptoms that overlap with those seen in COVID-19. It is therefore possible that medications targeting allergy may also ease symptoms from COVID-19.
Cytokines lead to systemic inflammation in viral illnesses
We know that certain cytokines e.g. IL-6, are known to contribute to lung and tissue inflammation, fever and fibrosis in response to viral infections (1). In patients who had died of SARS, elevated expressions of IL-1, IL-6 and TNF-α were found in infected lung and bronchial cells on autopsy (8). In influenza A, the accumulation and activation of mast cells in infected tissues is associated with local pathology (3).
Most hospitalised patients with COVID-19 have systemic elevation of pro-inflammatory cytokines e.g. IL-6 and TNF-α (7). In some cases these cytokines can exacerbate excessive inflammation especially in the lungs during COVID-19 (5). This is termed a “cytokine storm” in which mast cells play a central role (2). This can result in possible sepsis with fatal organ damage (3). The severity of pulmonary complications in COVID-19 is closely linked to IL-6 peak levels (9). The overactivation of mast cells and release of cytokines might also have a crucial role in the development of pulmonary fibrosis in COVID-19, particularly in populations pre-disposed to develop diseases related to mast cell activation (2). Pulmonary fibrosis may result in chronic lung disease in those who recover (10). Mast cell stabilisers may therefore have the potential to reduce mast cell cytokine release which may attenuate pulmonary complications, fatal inflammation and death in COVID-19 (2).
Improved survival in mice studies
Mast cell stabilisers have been shown to improve survival from sepsis in wild-type but not mast-cell deficient mice (11). In one study, all mice with viral-induced sepsis survived when given the mast cell stabiliser ketotifen with an antiviral; those given an antiviral or ketotifen in isolation had a lower survival rate (12). Ketotifen has also been shown to decrease lung lesions and cell death in mice infected with influenza A virus (12).
EVIDENCE OF MAST CELL STABILISERS, LEUKOTRIENE ANTAGONISTS AND ANTIHISTAMINES IN COVID-19
We searched PubMed and Google Scholar for studies that included terms for mast cell stabilisers, leukotriene antagonists or antihistamines and COVID-19. We did not find any studies that had examined the use of mast cell stabilisers, leukotriene antagonists or antihistamines in COVID-19. As our searches didn’t return any relevant results, we widened the criteria to include other acute viral respiratory tract infections.
EVIDENCE OF MAST CELL STABILISERS, LEUKOTRIENE ANTAGONISTS AND ANTIHISTAMINES IN ACUTE VIRAL RESPIRATORY TRACT INFECTION
Due to similarities of allergic upper respiratory symptoms with those of the common cold, studies have been undertaken to assess if anti-histamines have a role in alleviating symptoms in both conditions. A Cochrane review of 18 randomised controlled trials including adults and children (13) concluded that antihistamines have a limited short-term (days one and two of treatment) beneficial effect on severity of overall symptoms but not in the mid to long term. There was no clinically significant effect on nasal obstruction, rhinorrhoea or sneezing.
Excluding infant RSV (Respiratory Syncytial Virus) bronchiolitis, we did not find any relevant studies assessing the effect of beta agonists such as salbutamol (included in our search as a type of mast cell stabiliser), or leukotriene receptor antagonists such as montelukast, on clinical outcomes in acute viral respiratory tract infection.
Vitamin D has also been found to contribute to mast cell stabilisation (4). There is limited evidence that vitamin D supplementation over weeks to months is protective against acute respiratory tract infection in those individuals with low or very low vitamin D status (14). The evidence for treatment or prevention of COVID-19 with vitamin D is reviewed here (15).
CONCLUSIONS
- Severe pneumonia induced by human coronaviruses including SARS-CoV-2 is associated with raised pro-inflammatory cytokine responses, which result in acute pulmonary injury, severe acute respiratory syndrome and possible chronic pulmonary fibrosis. Mast cells are the main source of these pro-inflammatory cytokines and bronchoconstrictor mediators.
- At the time of writing, there was limited evidence supporting the use of antihistamines for short-term symptomatic relief of the common cold and none for the use of mast cell stabilisers, leukotriene antagonists or antihistamines in COVID-19.
- Clinical trials would be required to elucidate whether these drugs may be repurposed for the treatment of this disease. We suggest that it might be more appropriate to channel resources into pursuing alternative therapeutics that have a stronger evidence base for use in COVID-19 over those that are linked to mast cell stabilisation, cytokine modification or histamine release.
Disclaimer: this article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views stated here shuld not be used a substitute for professional medical advice.
Acknowledgements: We would like to thank Dr Jeffrey Aronson for his helpful comments and Dr Kilinç for kindly granting us permission to re-produce his diagram in Figure 1.
AUTHORS
Meriel Raymond* is a General Practitioner and an NIHR In-Practice Fellow based at the Nuffield Department of Primary Care Health Sciences, University of Oxford.
Gemma Ching-A-Sue* is a General Practitioner with an extended role in Ophthalmology at the John Radcliffe Hospital, Oxford.
Oliver van Hecke is a General Practitioner and an NIHR Academic Clinical Lecturer based at the Nuffield Department of Primary Care Health Sciences, University of Oxford.
* Meriel Raymond and Gemma Ching-A-Sue are co-first authors of this CEBM review.
SEARCH TERMS
We searched PubMed and Google Scholar on 22nd April 2020 using comprehensive terms to include the use of anti-histamines, mast cell stabilisers and leukotriene antagonists in any respiratory tract infection (see Table 1). The results were restricted to articles available in English. A more limited search was also run in LitCOVID and pre-prints sought from medRxiv.org. We also included reviews of the existing literature on this topic and reviewed relevant references from review articles. Both authors screened the titles and abstracts to select those relevant to this review, with advice from a third reviewer. We provide a narrative summary of the current literature.
Table 1. Search strategy
| Subgroup |
Expanded search terms used |
| Coronavirus |
2019nCoV, Wuhan coronavirus, Severe Acute Respiratory Syndrome Coronavirus 2, 2019-nCoV, WN-CoV, nCoV, SARS-CoV-2, HCoV-19, novel coronavirus |
| Respiratory infections |
SARS, MERS, pneumonia, acute respiratory infections, respiratory tract infections, influenza, common cold, bronchitis |
| Mast cell stabilisers |
Cromoglycate, ketotifen, nedocromil sodium, methylxanthine*, theophiline, aminophylline, Vitamin D, vit d*, colecalciferol, mast cell stab*, mastcell stab* beta 2 agonist*, b2 agonist*, salbutamol, terbutaline, salmeterol, bambuterol, formoterol |
| Leukotriene antagonists |
Montelukast |
| Antihistamines |
Antihistamine, hydroxyzine, chlorophenamine, acrivastine, blastine, cetirizine, fexofenadine, levocetirizine, loratidine, mizolastine, desloratidine, alimemazine, clemastine, cinnarizine |
REFERENCES
- Kritas SK, Ronconi G, Caraffa A, Gallenga CE, Ross R, Conti P. Mast cells contribute to coronavirus-induced inflammation: new anti-inflammatory strategy. J Biol Regul Homeost Agents [Internet]. 2020;34(1):2019. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32013309
- Taumann W, Molderings G. MC Sciences Statement – Additional COVID-19 Treatment Options [Internet]. 2020 [cited 2020 Apr 16]. Available from: https://www.mcsciences.com/news-2/
- Graham AC, Temple RM, Obar JJ. Mast cells and influenza A virus: Association with allergic responses and beyond. Front Immunol. 2015;6:1–12.
- Liu ZQ, Li XX, Qiu SQ, Yu Y, Li MG, Yang LT, et al. Vitamin D contributes to mast cell stabilization. Allergy Eur J Allergy Clin Immunol. 2017;72(8):1184–92.
- Kilinc E, Baranoglu Y. Mast cell stabilizers as a supportive therapy can contribute to alleviate fatal inflammatory responses and severity of pulmonary complications in COVID-19 infection. Anadolu Klin Tıp Bilim Derg. 2020;25(Supplement 1):111–8.
- NICE. Asthma: diagnosis, monitoring and chronic asthma management [Internet]. NICE guidance. 2017. Available from: www.nice.org.uk/guidance/ng80
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506.
- He L, Ding Y, Zhang Q, Che X, He Y, Shen H, et al. Expression of elevated levels of pro-inflammatory cytokines in SARS-CoV-infected ACE2+ cells in SARS patients: relation to the acute lung injury and pathogenesis of SARS. J Pathol. 2006 Nov;210(3):288–97.
- Russell B, Moss C, George G, Santaolalla A, Cope A, Papa S, et al. Associations between immune-suppressive and stimulating drugs and novel COVID-19—a systematic review of current evidence. ecancer. 2020;14:1022.
- Wang J, Wang BJ, Yang JC, Wang MY, Chen C, Luo GX, et al. Advances in the research of mechanism of pulmonary fibrosis induced by Corona Virus Disease 2019 and the corresponding therapeutic measures. Zhonghua Shao Shang Za Zhi. 2020;36:6.
- Ramos L, Peña G, Cai B, Deitch EA, Ulloa L. Mast Cell Stabilization Improves Survival by Preventing Apoptosis in Sepsis. J Immunol. 2010;185(1):709–16.
- Hu Y, Jin Y, Han D, Zhang G, Cao S, Xie J, et al. Mast Cell-Induced Lung Injury in Mice Infected with H5N1 Influenza Virus. J Virol. 2012;86(6):3347–56.
- De Sutter A, Saraswat A, Van Driel M. Antihistamines for the common cold ( Review ). Cochrane Database Syst Rev. 2015;(11).
- Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF, Bergman P, et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ. 2017;356.
- Lee J, Van Hecke O, Roberts N. Vitamin D: A rapid review of the evidence for treatment or prevention in COVID-19 [Internet]. The Centre for Evidence-Based Medicine, University of Oxford. 2020 [cited 2020 May 16]. Available from: https://www.cebm.net/covid-19/vitamin-d-a-rapid-review-of-the-evidence-for-treatment-or-prevention-in-covid-19/

