The accuracy of chest CT in the diagnosis of COVID-19: An umbrella review
March 4, 2021
Jae Young Park, Rosemary Freer, Richard Stevens, Neil Soneji, Nicholas Jones
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
Correspondence to nicholas.jones2@phc.ox.ac.uk
Verdict
- Among symptomatic adult patients, chest CT has a sensitivity for diagnosis of COVID-19 in excess of 90% but the specificity is lower, reportedly between 25 and 83%.
- Chest CT may have a role in the diagnosis of COVID-19 in a limited number of hospitalised patients, particularly where initial PCR testing has been inconclusive, or an alternative diagnosis is being considered.
- Where chest CT is used, bilateral changes with ground glass opacity with or without consolidation and interlobular septal thickening were the most common abnormalities among patients with COVID-19.
- Chest CT is infrequently used in children and we found only limited data regarding the diagnostic accuracy in COVID-19. However, based on this limited evidence the accuracy of chest CT appears lower among paediatric populations, suggesting it has a very limited role in these populations.
Background
The outbreak of COVID-19 caused by the SARS-CoV-2 virus began in Wuhan, China in December 2019 and has spread rapidly since. On March 12th 2020, the World Health Organisation (WHO) declared COVID-19 to be a pandemic.1 Up to the end of January 2021, there have been over 100 million confirmed cases of COVID-19 and over 2 million deaths, with the true figures likely to be higher.2
The diagnosis of COVID-19 currently relies largely on reverse transcriptase polymerase chain reaction (RT-PCR) testing of samples collected from the respiratory tract, most commonly through oro- or nasopharyngeal swabs. RT-PCR offers advantages in terms of low cost, safety and ease of collection. Initial challenges of access to RT-PCR testing kits have now been largely overcome and this is now a widely available technique. However, the sensitivity of RT-PCR testing ranges from 93% for samples collected at broncho-alveolar lavage to 63% for sputum and just 32% for throat swabs.3 Sensitivity may be lower if samples are collected sub-optimally or in patients with a low viral load. As a result, there are a significant number of false negative results, creating diagnostic uncertainty and the need for additional diagnostic tools to accurately differentiate between patients with and without COVID-19.4
Reports from regions with a high COVID-19 prevalence describe the use of chest computed tomography (CT) as one such diagnostic tool. Whilst guidelines in the United Kingdom (UK) do not advocate routine use of CT in COVID-19 diagnosis,5 6 the Royal College of Radiologists highlights that cases will arise where early CT imaging in patients with suspected COVID-19 will be clinically appropriate.5 This may be of particular relevance among hospital inpatients where there is a high index of suspicion that a patient has COVID-19 but a RT-PCR swab test is negative and other diagnostic tools, such as chest x-ray, are also indeterminate.4 7
Furthermore, patients with respiratory symptoms who do not have a confirmed diagnosis of COVID-19 may undergo CT for different indications, including diagnostic uncertainty and detection of complicating pulmonary thrombus. Thromboembolic disease is a common complication of COVID-19, with a recent meta-analysis reporting that the incidence of pulmonary embolism was 13% (95% CI:11 to 16%) among all hospitalised patients rising to 19% (95% CI:14 to 25%) among patients in Intensive Care Units (ITU), and most of these patients will have a CT pulmonary angiogram (CTPA) for diagnosis.8 It is important for radiologists to be aware of the key radiographic features of COVID-19 and if any can reliably differentiate COVID-19 from other lung pathology.
To evaluate the performance of chest CT in the diagnosis of COVID-19, we completed an umbrella review of published meta-analyses and systematic reviews. Our primary objective was to evaluate the diagnostic accuracy of chest CT for COVID-19 in both adults and children. Our secondary objectives were to identify the most common radiographic changes on chest CT among patients with COVID-19 and the distribution pattern of these changes within the lungs.
Methods
The review was completed as a rapid review without a pre-published protocol. The review question was designed around the following “PIRT” framework:
Population – patients with suspected COVID-19
Intervention – any protocol of a chest CT
Reference test – RT-PCR, though we also include other diagnostic modalities as determined by individual studies
Outcome – confirmed COVID-19
We searched PubMed, LitCOVID and MedRxiv, up to the 12th of May 2020 for literature reporting on the diagnostic accuracy of chest CT in COVID-19 patients, using keywords including “coronavirus”, “2019-nCoV”, “COVID-19”, “chest CT” and “CT Scan”. The full search strategy is included at the end of this document. Two reviewers (JYP and RF) independently screened titles and abstracts and selected articles for full-text review. The same two reviewers then independently assessed the full-text articles for inclusion in the review. Any discrepancies were resolved by discussion and consensus with a third reviewer (NJ).
Studies were eligible for inclusion if they were a meta-analysis that reported the sensitivity, specificity, and/or imaging details of a chest CT scan for diagnosis of COVID-19. We included meta-analyses of non-randomised studies as chest CT was predominantly done as part of routine clinical care and in addition to other diagnostic tests. Studies were excluded if they did not report key data such as population size. Case studies of individual patients were excluded. We excluded studies where chest CT was used to assess disease progression or for other non-diagnostic purposes, where CT diagnosis was made by artificial intelligence (AI) and not by a radiologist, reports which focused on the clinical features of COVID-19 without considering diagnostic accuracy, and studies or reports offering infection control guidance for radiologists. We excluded studies that only reported the localisation of a pre-specified subset of CT radiographic changes.
For each eligible meta-analysis, two investigators (JYP and RF) used a standardised template to extract information independently on first author, year of publication, outcome measures, number of included studies, total number of included cases, sensitivity, specificity, data describing the frequency of CT features, reference standard used for COVID-19 diagnosis, diagnostic criteria used for CT COVID-19 diagnosis, and whether the quality of included studies was assessed. These results were then cross checked against each other and combined.
For meta-analyses reporting both sensitivity and specificity we extracted data from the original studies included in these analyses to avoid double counting of results. From these primary studies we extracted data on the number of true positives, true negatives, false positives and false negatives for CT diagnosis of COVID-19 into a 2×2 table. We only included studies in this analysis that used RT-PCR as the reference standard.
These data were plotted on a single graph with receiver-operator characteristic axes.9 Bivariate meta-analysis of sensitivity and specificity was attempted but where this was not possible, univariate meta-analysis is reported instead. We also present univariate meta-analyses of reported CT features and localisation patterns. Meta-analyses were conducted with the inverse variance heterogeneity method, which uses the same weights as fixed effect inverse variance meta-analysis but allows for heterogeneity in the calculation of confidence intervals.10 We planned to report results based on study setting, whether patients were symptomatic for COVID-19 and key patient characteristics, such as age groups (adult versus child).
We performed a qualitative assessment of risk of bias across the included studies based upon the volume, quality, applicability and consistency of the evidence identified.
Results
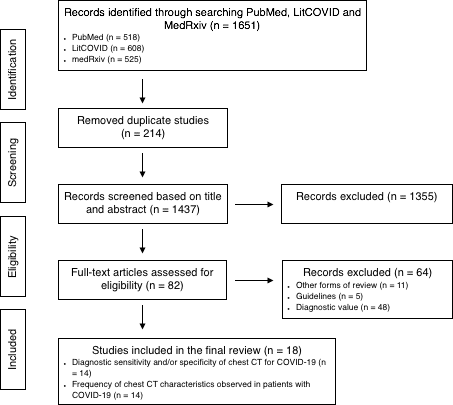
In total, 1651 articles were identified by the search strategy and 18 studies were ultimately included in the review (Figure 1).
Figure 1: Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram demonstrating the study selection process. CT = computed tomography.
Overview of included studies
Table 1 summarises the meta-analyses included in this umbrella review. Of these, 14 reported the sensitivity of chest CT for the diagnosis of COVID-19,11-24 10 of which included only adult patients,12-21 and three meta-analyses included paediatric patients.11 22 23 One meta-analysis reported data on both adult and paediatric patients.24 However, only 2 of these 14 studies also reported specificity, both of which included adult patients only (Table 1).21 24 Three of the studies reporting sensitivity results did so without reporting 95% confidence intervals.16 17 23
From these 14 meta-analyses, data was extracted from 44 non-overlapping primary studies for chest CT diagnostic performance, including 37 studies of adult patients and seven studies of children. Among these primary studies we identified nine that reported both sensitivity and specificity (or studies where it was possible to calculate this information) for chest CT.25-33
We further identified 14 meta-analyses that reported information on the frequency of chest CT radiographic features in patients with COVID-19.11 13-16 18-20 22 23 34-37 Of these, 11 included adults,13-16 18-20 34-37 and three meta-analyses included paediatric patients (Table 1).11 22 23 There were 12 meta-analyses that provided information on both sensitivity and specificity and CT characteristics.11-13,15-18,21-22, 24-26 From these, data were extracted from 122 primary studies reporting imaging findings on chest CT among patients with COVID-19 and 118 primary studies that reported the anatomical location of these changes.
Table 1: Details of the meta-analysis studies included in this review
| Author (Year) |
Journal |
Number of cases |
Reference Standard |
Sensitivity (%, 95%CI) |
Specificity (%, 95%CI) |
Localisation of CT features reported |
Quality assessment* |
| Adult Studies |
| Sun, P. et al. (2020)12 |
J Med Virol |
50466 |
Not stated |
0.97 (0.92 – 0.99) |
– |
No |
Yes |
| Bao, C. et al. (2020)13 |
J Am Coll Radiol |
2738 |
Not stated |
0.90 (0.84 – 0.94) |
– |
Yes |
No |
| Karimian, M. et al. (2020)14 |
medRxiv |
4183 |
RT-PCR and/or nucleic acid testing |
0.95 (0.92 – 0.96) |
– |
Yes |
Yes |
| Lv, M. et al. (2020)15 |
Ann Transl Med |
5694 |
RT-PCR |
0.99 (0.97 – 1.00) |
– |
Yes |
Yes |
| Fang, Z. et al. (2020)17 |
medRxiv |
2866 |
RT-PCR or coronavirus antibody detection |
0.88 |
– |
No |
Yes |
| Heydari, K. et al. (2020)18 |
medRxiv |
49504 |
Described as “Positive for pneumonia”, presumed clinical diagnosis |
0.84 (0.79 – 0.88) |
– |
Yes |
Yes |
| Zhu, J. et al. (2020)20 |
J Med Virol |
4121 |
Not stated |
0.92 (0.86- 0.96) |
– |
Yes |
Yes |
| Venugopal, V. et al. (2020)16 |
medRxiv |
3615 |
RT-PCR |
0.93 |
– |
Yes |
Yes |
| Qian, Q. et al. (2020)19 |
medRxiv |
356 |
Not stated |
0.89 (0.80 – 0.94) |
– |
Yes |
Yes |
| Salehi, S. et al. (2020)34 |
AJR Am J Roentgenol |
919 |
RT-PCR |
– |
– |
Yes |
Yes |
| Fu. L. et al. (2020)35 |
J Infect |
3600 |
RT-PCR |
– |
– |
Yes |
Yes |
| Vaseghi, G. et al. (2020)36 |
medRxiv |
293 |
RT-PCR |
– |
– |
Yes |
Yes |
| Mao, Y, et al. (2020)37 |
medRxiv |
47936 |
Not-stated |
– |
–
|
Yes |
No |
| Huang, E. et al. (2020)21 |
Crit Care |
1286 |
RT-PCR |
0.95 (0.93 – 0.97) |
0.09 (0.02 – 0.34) |
No |
No |
| Paediatric studies |
| de Souza. et al. (2020)23 |
Paediat Pulmon |
184 |
RT-PCR |
0.63 |
– |
Yes |
No |
| Wang, Z. et al. (2020)22 |
Ann Trans Med |
1667 |
Not-stated |
0.66 (0.55 – 0.77) |
– |
Yes |
Yes |
| Chang, T. et al. (2020)11 |
J Formos Med Assoc |
52 |
RT-PCR |
0.93 (0.57 – 0.82) |
– |
Yes |
Yes |
| Studies including adults and children |
| Kim, H. et al. (2020)24 |
Radiology |
6218 |
RT-PCR |
0.94 (0.91 – 0.96) |
0.37 (0.26 – 0.50) |
No |
Yes |
Abbreviations: CT = computed tomography; RT-PCR = reverse transcriptase polymerase chain reaction
*NB – results have been rounded to two decimal places
Quality of evidence
There was heterogeneity between studies in terms of study setting, included participants and reference test. Whilst RT-PCR was the most common reference standard, the site of collection for the sample was frequently not reported. Furthermore, one primary study used a clinical diagnosis of pneumonia as the reference standard and many others were not explicit about the reference standard used (Table 1). The meta-analyses we reviewed provided limited information as to the original study setting or context in which the CT scan was being done, nor information on patient characteristics such as co-morbid disease, nor information on SARS-CoV-2 viral load, symptoms, or number of days since infection began (Table 2). Among 37 primary studies reporting sensitivity for chest CT in the diagnosis of COVID-19, four studies had a majority of asymptomatic patients,38-41 while the remaining studies either had a majority of symptomatic patients or all symptomatic patients. However, reporting of symptoms was also inconsistent and was not always explicit in individual studies.
Four of the meta-analyses included did not conduct any type of quality assessment of the studies in their review. The sample size of the primary studies tended to be small, with many including less than 100 participants (Figure 2). Inclusion criteria in the primary studies were not always explicit but often relied on convenience sampling or retrospective analysis of CT results, where there is a risk of selection bias given those with greater symptom burden may be more likely to receive a CT scan. Despite these limitations there was consistency between the nine primary studies regarding the high sensitivity and relatively low specificity of CT chest for diagnosing COVID-19.
Table 2: Summary of the body of evidence
| |
Description |
| Volume |
A large volume of observational data was identified, including 18 meta-analyses and 44 non-overlapping primary studies reporting the diagnostic performance of chest CT for COVID-19, 122 primary studies reporting imaging findings on chest CT among patients with COVID-19 and 118 primary studies reporting the distribution of these changes. |
| Quality |
Although we include 18 meta-analyses, these were based on a heterogeneous group of primary studies with frequent key limitations. The tests used as a reference standard were inconsistent. RT-PCR was commonly used but collected from different body sites, which is known to affect diagnostic accuracy. Study setting (e.g. Accident and Emergency versus hospitalised patients versus ITU) or disease severity were rarely reported. Baseline population characteristics such as co-morbidities and smoking status were also infrequently reported. The number of days a patient had been symptomatic and viral load were not reported yet may be relevant when interpreting test results. |
| Applicability
|
The majority of studies were conducted in China, reflecting where the first confirmed outbreak of COVID-19 took place. Strict contact tracing of symptomatic patients was conducted in China, meaning asymptomatic patients were detected with COVID-19 and included in some studies, which is likely to impact on the diagnostic accuracy of chest CT. A subgroup analysis was undertaken excluding these studies. |
| Consistency |
Overall, there was consistency among the meta-analyses and primary studies that chest CT offers high sensitivity but relatively low specificity for the diagnosis of COVID-19 among adult patients. Studies of the sensitivity of chest CT for diagnosing COVID-19 in children were less consistent, ranging from moderate to poor. |
Diagnostic performance of chest CT scan
Bivariate meta-analysis of sensitivity and specificity was attempted but models did not converge. Inspection of the data suggested this was due to high heterogeneity of specificity, while sensitivity was relatively constant. We therefore present univariate meta-analysis of sensitivity but did not attempt to pool specificity estimates.
In adults, the reported sensitivities of CT for COVID-19 diagnosis with RT-PCR reference standard ranged from 61% to 100% (Figure 2) across 37 individual studies25-29 31-33 38-40 42-67. The pooled sensitivity for studies including symptomatic patients or where the majority of patients were symptomatic was 94.4% (95% CI: 90.3 to 98.5, I2=92.3%). There was substantial heterogeneity, which may reflect important differences in study population and methods, including setting, timing of CT in disease course and CT protocols. For example, the largest study of symptomatic patients (n=1,099) included hospitalised patients with ‘severe’ disease (n=173), but also outpatients with ‘non-severe’ symptoms (n=926).68 Other primary studies, including the second largest, did not report data on study setting or disease severity, precluding a subgroup analysis on this criteria.25
The sensitivity of chest CT tended to be lower among studies that included mostly or entirely asymptomatic patients. We found four such studies and in a pooled analysis, the sensitivity for COVID-19 diagnosis was 66.1% (95% CI: 60.4 to 71.8%, I2=0.0%). This represents a statistically significant difference between the sensitivity of chest CT among symptomatic patients compared to those who are asymptomatic.
In addition, we found two meta-analyses of paediatric populations, which both reported lower sensitivity results when compared to adult populations (66.0%, 95% CI: 55.0 to 77.0 and 73.0%, 95% CI: 57.0 to 82.0 respectively). There was substantial heterogeneity in both datasets. Among primary studies, the sensitivity of chest CT in children ranged from 0% to 80%.69-75
Figure 2: Summary data for the individual studies of CT sensitivity in COVID-19 patients

We identified nine primary studies that reported both sensitivity and specificity data, all in adult patients using RT-PCR as the reference standard (Figure 3, Table 1).25-30 32 33 40 The reported sensitivity of chest CT for COVID-19 diagnosis ranged from 61% to 100%. The specificity ranged from 25% to 83%. The sensitivity and specificity reported in the primary studies are plotted in ROC space in Figure 3.
Figure 3: Sensitivity and specificity of CT chest for the diagnosis of COVID-19 in adults across five primary studies that reported both.

Studies are labelled as follows; A: Ai et al,25 L: Li et al,26 w: Wang et al,27 X: Xie et al,33 Z: Zhu et al,28 C: Caruso et al,29 H: Himoto et al,32 a: Ai et al,30 W: Wei et a.l31
Radiographic features on chest CT scan among patients with COVID-19
The most frequently observed radiographic feature was ground glass opacity (GGO) both in adults (0.66, 95% CI: 0.62 to 0.69) and children (0.39, 95% CI: 0.31 to 0.46) (Table 3). The other most frequently reported abnormalities in adults were interlobular septal thickening (0.65, 95% CI: 0.58 to 0.71) and consolidation with GGO (0.51, 95% CI: 0.46 to 0.57). Of note, some of the reported radiographic features are incidental findings or may be overlapping between different studies, where studies used different terminology to describe similar radiographic findings.
Among adults, chest CT changes were most frequently found in the right lower lobe (0.87, 95% CI: 0.81 to 0.93) but were also commonly found in the left lower lobe (0.81, 95% CI: 0.81 to 0.93), left upper lobe (0.69, 95% CI: 0.59 to 0.78) and right upper lobe (0.64, 95% CI: 0.55 to 0.73). CT changes also tended to be peripheral, (0.74, 95% CI: 0.63 to 0.81), bilateral (0.81, 95% CI: 0.78 to 0.83) and/or multi-lobular (0.67, 95% CI: 0.55 to 0.79). Only limited data for paediatric patients was available, but in our pooled analysis 28% of children had unilateral chest CT changes (0.28, 95% CI: 0.20 to 0.36) and 31% bilateral changes (0.31 (95% CI: 0.19 to 0.43).
Table 3: Pooled prevalence of features described on chest CT in patients with COVID-19, reported in order of frequency detected.
| Features |
Pooled prevalence in adults |
Pooled prevalence in children |
| Ground glass opacity |
0.66 (95% CI: 0.62, 0.69) |
0.39 (95% CI: 0.31, 0.46) |
| Interlobular septal thickening |
0.65 (95% CI: 0.58, 0.71) |
– |
| Ground glass opacity with consolidation |
0.51 (95% CI: 0.46, 0.57) |
– |
| Air bronchograms |
0.49 (95% CI: 0.40, 0.57) |
– |
| Consolidation |
0.35 (95% CI: 0.29, 0.40) |
0.31 (95% CI: 0.10, 0.52) |
| Pleural thickening |
0.30 (95% CI: 0.20, 0.39) |
– |
| Fibrotic changes |
0.17 (95% CI: -0.02, 0.37) |
– |
| Reticular findings* |
0.17 (95% CI: -0.06, 0.41) |
– |
| Crazy paving patterns* |
0.16 (95% CI: 0.12, 0.19) |
– |
| Pleural effusion |
0.06 (95% CI: 0.05, 0.07) |
– |
| Pericardial effusion |
0.05 (95% CI: 0.02, 0.07) |
– |
| Lymphadenopathy |
0.04 (95% CI: 0.03, 0.06) |
– |
There may be overlap in findings between these categories, reflecting differences in the terminology used in the original studies. Common incidental findings are also reported. For example, reticular findings* may also be defined as septal thickening in other included studies. Similarly, crazy paving patterns* may be used to describe ground class opacity with septal thickening. For each category the definition used reflect those in the original studies.
Discussion
In this umbrella review, we found that CT chest has a high sensitivity but lower specificity for the diagnosis of COVID-19. The sensitivity of chest CT was higher among symptomatic patients compared to studies where the majority of patients were asymptomatic. Among nine primary studies of adult patients that reported both specificity and sensitivity using RT-PCR as a reference standard, sensitivity ranged from 61% to 100% and specificity ranged from 25% to 83%. In children, sensitivity of chest CT dropped to approximately 70%, and no reports of specificity were identified.
A multitude of radiographic changes on chest CT were reported among patients with COVID-19, but the most prevalent among adults were GGO (0.66, 95% CI: 0.62 to 0.69), interlobular septal thickening (0.65, 95% CI: 0.58 to 0.71) and consolidation with GGO (0.51, 95% CI: 0.46 to 0.57). Where present, radiographic changes were frequently in the lower lobes, peripheral and were also bilateral in over 80% of cases (0.81, 95% CI: 0.78 to 0.83) and/or multi-lobular (0.67, 95% CI: 0.55 to 0.79). These findings may help radiologists when considering whether a chest CT with positive findings is supportive of a diagnosis of COVID-19.
Comparison to existing literature
Our findings are consistent with previous meta-analyses that report that a diagnosis of COVID-19 is less likely among patients with a normal chest CT. For example, a recently published systematic review and meta-analysis of chest CT accuracy in COVID-19 diagnosis reported the sensitivity and specificity of chest CT were 87% (95% CI: 85 to 90%) and 43% (95% CI: 29 to 63%) respectively, when considering studies that used RT-PCR as a reference standard.76 Our study is the first to use SROC analysis to plot comparative sensitivity and specificity results across studies but supports the recommendations of this review that CT chest should be seen as an adjuvant diagnostic test only.
Although there is now extensive reporting on the CT features of COVID-19, distinguishing COVID-19 from other viral pneumonia remains a challenge and limits specificity. The most frequently reported CT features in patients with COVID-19 including GGO are also commonly found in other viral pneumonias.77 78 Similarly, the lower lobe and bilateral predominance of radiographic changes are also common patterns of distribution in other viral pneumonias.49 Furthermore, many patients hospitalised with COVID-19 will go on to develop further lung pathology, including acute respiratory distress syndrome (ARDS) or thromboembolic disease, which may complicate the interpretation of CT imaging. Whilst our results help to identify expected findings on chest CT, diagnosis should be based on virology results, clinical suspicion, population prevalence of COVID-19 and other common viral pathogens at the time. Further studies could aid CT diagnosis by further elucidating key features that distinguish COVID-19 infection from other viral pneumonias.
Strengths and limitations
To our knowledge, this is the first umbrella review of meta-analyses in this field to date. We include data from 18 meta-analyses and extracted data from over 150 primary studies allowing us to pool sensitivity results without the risk of overlapping data. Nonetheless, there are key limitations in the evidence base to date that are important to recognise. Studies were observational and conducted across a range of settings, including both symptomatic and asymptomatic patients. Limited information is given regarding the role of CT in determining the COVID-19 diagnosis, and with regard to important patient characteristics such as days since admission or infection, viral load, severity of illness or underlying co-morbidities, which all may determine chest CT findings in this context. For example, fibrotic changes may be a late feature of COVID-19, but could also be an incidental finding, particularly in the acute phase. Many studies included fewer than 100 participants and may be at risk of selection bias, with the sickest patients most likely to undergo imaging. Whilst there is reasonable consistency across the studies in terms of sensitivity and specificity, the stated limitations suggest further research is needed to understand the optimal role of chest CT dependent on patient and disease characteristics.
Lower sensitivity of CT for COVID-19 diagnosis in children may be reflect the milder symptoms and typically more benign disease course in this population.79 80 Due to a milder disease course and a higher risk associated with radiation exposure in children, CT is rarely used for the diagnosis of COVID-19 in the pediatric population. Consequently only 3 meta-analyses were identified that reported on CT use in children with COVID-19. This relative paucity of available data may limit the reliability of estimates for CT sensitivity in pediatric populations.
Implications for practice
Our results support the published guidance by The Royal College of Radiologists that chest CT should not be used alone to diagnose COVID-19.5 Beyond limitations in the diagnostic accuracy of chest CT, there are barriers to use in practice such as scanning capacity, which is further reduced due to time allocations for decontamination following imaging. CT is also comparatively expensive and results in higher radiation exposure for patients.
Despite this, chest CT may have a role in the diagnostic assessment of some patients with suspected COVID-19. Up to 25% of patients with COVID-19 may have false-negative RT-PCR results, particularly if samples are collected incorrectly or where patients are in the early stages of disease and may have a low viral load.61 A recent report from the Royal Free Hospital in London advocates the use of chest CT in patients who have a high clinical suspicion of COVID-19 but who have had two negative RT-PCR tests and a negative or indeterminate chest x-ray.4 Our results suggest that in this context, a negative chest CT scan could help to rule out COVID-19 and should prompt clinicians to consider alternative diagnoses. However, a normal chest CT scan alone should not be used to exclude COVID-19.
COVID-19 may also be seen as an incidental and unexpected finding in asymptomatic patients undergoing a CT chest for other reasons, such as oncology follow-up. In the UK, radiologists are currently recommended to report on the likelihood of COVID-19 for all chest CT scans, with the British Society of Thoracic Imaging suggesting results should be graded as ‘classic’ (“100% confidence for COVID), ‘probable’ (71-99% percent confidence for COVID), ‘indeterminate’ (<70% confidence for COVID) or ‘non-COVID’ (70% confidence for alternative) based on set descriptions. As such, it is important that radiologists are aware of key diagnostic features of the disease on chest CT. Our results suggest GGO (with or without consolidation) and interlobular septal thickening, particularly where bilateral or in the lower lobes are supportive of a diagnosis of COVID-19. However, given the frequency with which these changes are seen in other chest pathology, radiologists and clinicians should work together to contextualise such findings based on the individual case presentation, particularly given our finding suggesting the sensitivity of chest CT is significantly lower in asymptomatic patients.
CONCLUSIONS
- Among symptomatic adult patients, chest CT has a sensitivity for diagnosis of COVID-19 in excess of 90% but the specificity is lower, reportedly between 25 and 83%.
- Chest CT may have a role in the diagnosis of COVID-19 in a limited number of hospitalised patients, particularly where initial PCR testing has been inconclusive, or an alternative diagnosis is being considered.
- Where chest CT is used, bilateral changes with ground glass opacity with or without consolidation and interlobular septal thickening were the most common abnormalities among patients with COVID-19.
- Chest CT is infrequently used in children and we found only limited data regarding the diagnostic accuracy in COVID-19. However, based on this limited evidence the accuracy of chest CT appears lower among paediatric populations, suggesting it has a very limited role in these populations.
Disclaimer: the article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.
AUTHORS
Jae Park is a medical student at the University of Oxford.
Rosemary Freer is a medical student at the University of Oxford.
Richard Stevens is an Associate Professor in Medical Statistics and the Deputy Director of the Statistics Group in the Nuffield Department of Primary Care Health Sciences, University of Oxford.
Neil Soneji is a Consultant Radiologist at Imperial College Healthcare NHS Trust and the Royal Marsden Hospital NHS Foundation Trust, London UK
Nicholas Jones is a GP and Wellcome Trust Doctoral Research Fellow at the Nuffield Department of Primary Care Health Sciences, University of Oxford.
FUNDING
No direct funding was received for this review. Nicholas Jones is supported by a Wellcome Trust Doctoral Research Fellowship (203921/Z/16/Z)
SEARCH TERMS
| Search portal
|
Algorithm
|
Number of papers identified |
| PubMed |
Search: ((coronavirus*[Title] OR coronovirus*[Title] OR coronoravirus*[Title] OR coronaravirus*[Title] OR corono-virus*[Title] OR corona-virus*[Title] OR “Coronavirus”[Mesh] OR “Coronavirus Infections”[Mesh] OR “Wuhan coronavirus” [Supplementary Concept] OR “Severe Acute Respiratory Syndrome Coronavirus 2″[Supplementary Concept] OR COVID-19[All Fields] OR CORVID-19[All Fields] OR “2019nCoV”[All Fields] OR “2019-nCoV”[All Fields] OR WN-CoV[All Fields] OR nCoV[All Fields] OR “SARS-CoV-2”[All Fields] OR HCoV-19[All Fields] OR “novel coronavirus”[All Fields])) AND ((“Tomography, X-Ray Computed”[Mesh]) OR ((computed tomogra*[Title/Abstract] OR “ct chest”[Title/Abstract] OR “chest ct”[Title/Abstract] OR ct scan*[Title/Abstract]) OR (ct[Title]))) Filters: from 2020 – 2020 |
518 |
| LitCOVID |
computed tomogra* OR “ct chest” OR “chest ct” OR ct scan* OR ct[ti] |
608 |
| medRxiv |
“computed tomography” (match phrase words) and abstract or title “coronavirus covid-19” (match any words) |
193 |
| medRxiv |
title “chest ct” (match phrase words) and abstract or title “coronavirus covid-19” |
321 |
| medRxiv |
“ct chest” (match phrase words) and abstract or title “coronavirus covid-19” |
11 |
| Total number of papers identified |
1651 |
REFERENCESPark
- World Health Organization. WHO/Europe | Coronavirus disease (COVID-19) outbreak – WHO announces COVID-19 outbreak a pandemic https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/news/news/2020/3/who-announces-covid-19-outbreak-a-pandemic2020 [accessed 23 June 2020.
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard https://covid19.who.int/2021 [accessed 26 January 2021.
- Watson J, Whiting PF, Brush JE. Interpreting a covid-19 test result. BMJ 2020;369:m1808. doi: 10.1136/bmj.m1808 [published Online First: 2020/05/14]
- Tavare AN, Braddy A, Brill S, et al. Managing high clinical suspicion COVID-19 inpatients with negative RT-PCR: a pragmatic and limited role for thoracic CT. Thorax 2020;75(7):537-38. doi: 10.1136/thoraxjnl-2020-214916 [published Online First: 2020/04/23]
- The Royal College of Radiologists. The role of CT in patients suspected with COVID-19 infection https://www.rcr.ac.uk/college/coronavirus-covid-19-what-rcr-doing/clinical-information/role-ct-chest/role-ct-patients 2020 [accessed 23 June 2020.
- Society of Thoracic Radiology. Society of Thoracic Radiology and The American Society of Emergency Radiology COVID-19 Position Statement https://thoracicrad.org/wp-content/uploads/2020/03/STR-ASER-Position-Statement-1.pdf2020 [accessed 23 June 2020.
- Liu J, Yu H, Zhang S. The indispensable role of chest CT in the detection of coronavirus disease 2019 (COVID-19). Eur J Nucl Med Mol Imaging 2020;47(7):1638-39. doi: 10.1007/s00259-020-04795-x [published Online First: 2020/04/05]
- Malas MB, Naazie IN, Elsayed N, et al. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. EClinicalMedicine 2020;29:100639. doi: 10.1016/j.eclinm.2020.100639 [published Online First: 2020/12/01]
- Hajian-Tilaki K. Receiver Operating Characteristic (ROC) Curve Analysis for Medical Diagnostic Test Evaluation. Caspian J Intern Med 2013;4(2):627-35. [published Online First: 2013/09/07]
- Doi SA, Barendregt JJ, Khan S, et al. Advances in the meta-analysis of heterogeneous clinical trials I: The inverse variance heterogeneity model. Contemp Clin Trials 2015;45(Pt A):130-8. doi: 10.1016/j.cct.2015.05.009 [published Online First: 2015/05/25]
- Chang TH, Wu JL, Chang LY. Clinical characteristics and diagnostic challenges of pediatric COVID-19: A systematic review and meta-analysis. J Formos Med Assoc 2020;119(5):982-89. doi: 10.1016/j.jfma.2020.04.007 [published Online First: 2020/04/21]
- Sun P, Qie S, Liu Z, et al. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: A single arm meta-analysis. J Med Virol 2020;92(6):612-17. doi: 10.1002/jmv.25735 [published Online First: 2020/02/29]
- Bao C, Liu X, Zhang H, et al. Coronavirus Disease 2019 (COVID-19) CT Findings: A Systematic Review and Meta-analysis. J Am Coll Radiol 2020;17(6):701-09. doi: 10.1016/j.jacr.2020.03.006 [published Online First: 2020/04/14]
- Karimian M, Azami M. Chest computed tomography (CT) scan findings in patients with COVID-19: A systematic review and meta-analysis. medRxiv 2020 doi: 10.1101/2020.04.22.20075382 [published Online First: April 25, 2020]
- Lv M, Wang M, Yang N, et al. Chest computed tomography for the diagnosis of patients with coronavirus disease 2019 (COVID-19): a rapid review and meta-analysis. Ann Transl Med 2020;8(10):622. doi: 10.21037/atm-20-3311 [published Online First: 2020/06/23]
- Venugopal VK, Mahajan V, Rajan S, et al. A systematic meta-analysis of CT features of COVID-19: Lessons from radiology. medRxiv 2020 doi: 10.1101/2020.04.04.20052241 [published Online First: April 7, 2020]
- Fang Z, Yi F, Wu K, et al. Clinical Characteristics of Coronavirus Disease 2019 (COVID-19): An Updated Systematic Review medRxiv 2020 doi: 10.1101/2020.03.07.20032573 [published Online First: March 12, 2020]
- Heydari K, Rismantab S, Shamshirian A, et al. Clinical and Paraclinical Characteristics of COVID-19 patients: A systematic review and meta-analysis. medRxiv 2020 doi: 10.1101/2020.03.26.20044057 [published Online First: March 30, 2020]
- Qian K, Deng Y, Tai Y, et al. Clinical Characteristics of 2019 Novel Infected Coronavirus Pneumonia:A Systemic Review and Meta-analysis. . medRxiv 2020 doi: 10.1101/2020.02.14.20021535 [published Online First: February 17, 2020]
- Zhu J, Zhong Z, Li H, et al. CT imaging features of 4121 patients with COVID‐19: A meta‐analysis. Journal of Medical Virology 2020 doi: 10.1002/jmv.25910 [published Online First: 21 April 2020 ]
- Huang EP, Sung CW, Chen CH, et al. Can computed tomography be a primary tool for COVID-19 detection? Evidence appraisal through meta-analysis. Crit Care 2020;24(1):193. doi: 10.1186/s13054-020-02908-4 [published Online First: 2020/05/08]
- Wang Z, Zhou Q, Wang C, et al. Clinical characteristics of children with COVID-19: a rapid review and meta-analysis. Ann Transl Med 2020;8(10):620. doi: 10.21037/atm-20-3302 [published Online First: 2020/06/23]
- de Souza TH, Nadal JA, Nogueira RJN, et al. Clinical manifestations of children with COVID-19: A systematic review. Pediatr Pulmonol 2020;55(8):1892-99. doi: 10.1002/ppul.24885 [published Online First: 2020/06/04]
- Kim H, Hong H, Yoon SH. Diagnostic Performance of CT and Reverse Transcriptase Polymerase Chain Reaction for Coronavirus Disease 2019: A Meta-Analysis. Radiology 2020;296(3):E145-E55. doi: 10.1148/radiol.2020201343 [published Online First: 2020/04/18]
- Ai T, Yang Z, Hou H, et al. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology 2020;296(2):E32-E40. doi: 10.1148/radiol.2020200642 [published Online First: 2020/02/27]
- Li K, Wu J, Wu F, et al. The Clinical and Chest CT Features Associated With Severe and Critical COVID-19 Pneumonia. Invest Radiol 2020;55(6):327-31. doi: 10.1097/RLI.0000000000000672 [published Online First: 2020/03/03]
- Wang L, Gao YH, Lou LL, et al. The clinical dynamics of 18 cases of COVID-19 outside of Wuhan, China. Eur Respir J 2020;55(4) doi: 10.1183/13993003.00398-2020 [published Online First: 2020/03/07]
- Zhu W, Xie K, Lu H, et al. Initial clinical features of suspected coronavirus disease 2019 in two emergency departments outside of Hubei, China. J Med Virol 2020;92(9):1525-32. doi: 10.1002/jmv.25763 [published Online First: 2020/03/14]
- Caruso D, Zerunian M, Polici M, et al. Chest CT Features of COVID-19 in Rome, Italy. Radiology 2020;296(2):E79-E85. doi: 10.1148/radiol.2020201237 [published Online First: 2020/04/04]
- Ai J, Zhang H, Xu T, et al. Optimizing diagnostic strategy for novel coronavirus pneumonia, a multi-center study in Eastern China. medRxiv 2020 doi: 10.1101/2020.02.13.20022673 [published Online First: February 17, 2020]
- Wei Y, Lu Y, Xia L, et al. Analysis of 2019 novel coronavirus infection and clinical characteristics of outpatients: An epidemiological study from a fever clinic in Wuhan, China. J Med Virol 2020;92(11):2758-67. doi: 10.1002/jmv.26175 [published Online First: 2020/06/17]
- Himoto Y, Sakata A, Kirita M, et al. Diagnostic performance of chest CT to differentiate COVID-19 pneumonia in non-high-epidemic area in Japan. Jpn J Radiol 2020;38(5):400-06. doi: 10.1007/s11604-020-00958-w [published Online First: 2020/04/02]
- Xie C, Jiang L, Huang G, et al. Comparison of different samples for 2019 novel coronavirus detection by nucleic acid amplification tests. Int J Infect Dis 2020;93:264-67. doi: 10.1016/j.ijid.2020.02.050 [published Online First: 2020/03/03]
- Salehi S, Abedi A, Balakrishnan S, et al. Coronavirus Disease 2019 (COVID-19): A Systematic Review of Imaging Findings in 919 Patients. AJR Am J Roentgenol 2020;215(1):87-93. doi: 10.2214/AJR.20.23034 [published Online First: 2020/03/17]
- Fu L, Wang B, Yuan T, et al. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J Infect 2020;80(6):656-65. doi: 10.1016/j.jinf.2020.03.041 [published Online First: 2020/04/14]
- Vaseghi G, Mansourian M, Karimi R, et al. Clinical characterization and chest CT findings in laboratory-confirmed COVID-19: A systematic review and meta-analysis. medRxiv 2020 doi: 10.1101/2020.03.05.20031518 [published Online First: March 8, 2020]
- Mao Y, Lin W, Wen J, et al. Clinical and pathological characteristics of 2019 novel coronavirus disease (COVID-19): A systematic reviews. medRxiv 2020 doi: 10.1101/2020.02.20.20025601 [published Online First: March 19, 2020]
- Lei Y, Huang X, SiLang B, et al. Clinical features of imported cases of coronavirus disease 2019 in Tibetan patients in the Plateau area. medRxiv 2020:2020.03.09.20033126. doi: 10.1101/2020.03.09.20033126
- Inui S, Fujikawa A, Jitsu M, et al. Chest CT Findings in Cases from the Cruise Ship “Diamond Princess” with Coronavirus Disease 2019 (COVID-19). Radiol Cardiothorac Imaging 2020;2(2):e200110. doi: 10.1148/ryct.2020200110
- Wang Y, Liu Y, Liu L, et al. Clinical Outcomes in 55 Patients With Severe Acute Respiratory Syndrome Coronavirus 2 Who Were Asymptomatic at Hospital Admission in Shenzhen, China. J Infect Dis 2020;221(11):1770-74. doi: 10.1093/infdis/jiaa119 [published Online First: 2020/03/18]
- Hu Z, Song C, Xu C, et al. Clinical characteristics of 24 asymptomatic infections with COVID-19 screened among close contacts in Nanjing, China. Sci China Life Sci 2020;63(5):706-11. doi: 10.1007/s11427-020-1661-4 [published Online First: 2020/03/09]
- Ai J-W, Zhang H-C, Xu T, et al. Optimizing diagnostic strategy for novel coronavirus pneumonia, a multi-center study in Eastern China. medRxiv 2020:2020.02.13.20022673. doi: 10.1101/2020.02.13.20022673
- Cai Q, Huang D, Ou P, et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy 2020;75(7):1742-52. doi: 10.1111/all.14309 [published Online First: 2020/04/03]
- Chen J, Qi T, Liu L, et al. Clinical progression of patients with COVID-19 in Shanghai, China. J Infect 2020;80(5):e1-e6. doi: 10.1016/j.jinf.2020.03.004 [published Online First: 2020/03/17]
- Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020;395(10223):507-13. doi: 10.1016/S0140-6736(20)30211-7 [published Online First: 2020/02/03]
- Chen Z, Fan H, Cai J, et al. High-resolution computed tomography manifestations of COVID-19 infections in patients of different ages. Eur J Radiol 2020;126:108972. doi: 10.1016/j.ejrad.2020.108972 [published Online First: 2020/04/03]
- Fang Y, Zhang H, Xie J, et al. Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020;296(2):E115-E17. doi: 10.1148/radiol.2020200432 [published Online First: 2020/02/20]
- Fu H, Li H, Tang X, et al. Analysis on the Clinical Characteristics of 36 Cases of Novel Coronavirus Pneumonia in Kunming. medRxiv 2020:2020.02.28.20029173. doi: 10.1101/2020.02.28.20029173
- Guan CS, Lv ZB, Yan S, et al. Imaging Features of Coronavirus disease 2019 (COVID-19): Evaluation on Thin-Section CT. Acad Radiol 2020;27(5):609-13. doi: 10.1016/j.acra.2020.03.002 [published Online First: 2020/03/25]
- Guan W-j, Ni Z-y, Hu Y, et al. Clinical characteristics of 2019 novel coronavirus infection in China. medRxiv 2020:2020.02.06.20020974. doi: 10.1101/2020.02.06.20020974
- Hu Z, Song C, Xu C, et al. Clinical characteristics of 24 asymptomatic infections with COVID-19 screened among close contacts in Nanjing, China. Science China Life Sciences 2020;63(5):706-11. doi: 10.1007/s11427-020-1661-4
- Huang G, Gong T, Wang G, et al. Timely Diagnosis and Treatment Shortens the Time to Resolution of Coronavirus Disease (COVID-19) Pneumonia and Lowers the Highest and Last CT Scores From Sequential Chest CT. AJR Am J Roentgenol 2020;215(2):367-73. doi: 10.2214/AJR.20.23078 [published Online First: 2020/04/01]
- Li K, Fang Y, Li W, et al. CT image visual quantitative evaluation and clinical classification of coronavirus disease (COVID-19). Eur Radiol 2020;30(8):4407-16. doi: 10.1007/s00330-020-06817-6 [published Online First: 2020/03/28]
- Ling Z, Xu X, Gan Q, et al. Asymptomatic SARS-CoV-2 infected patients with persistent negative CT findings. Eur J Radiol 2020;126:108956. doi: 10.1016/j.ejrad.2020.108956 [published Online First: 2020/03/22]
- Liu J, Ouyang L, Guo P, et al. Epidemiological, Clinical Characteristics and Outcome of Medical Staff Infected with COVID-19 in Wuhan, China: A Retrospective Case Series Analysis. medRxiv 2020:2020.03.09.20033118. doi: 10.1101/2020.03.09.20033118
- Liu K, Chen Y, Lin R, et al. Clinical features of COVID-19 in elderly patients: A comparison with young and middle-aged patients. J Infect 2020;80(6):e14-e18. doi: 10.1016/j.jinf.2020.03.005 [published Online First: 2020/03/17]
- Long C, Xu H, Shen Q, et al. Diagnosis of the Coronavirus disease (COVID-19): rRT-PCR or CT? Eur J Radiol 2020;126:108961. doi: 10.1016/j.ejrad.2020.108961 [published Online First: 2020/04/02]
- Wang K, Kang S, Tian R, et al. Imaging manifestations and diagnostic value of chest CT of coronavirus disease 2019 (COVID-19) in the Xiaogan area. Clin Radiol 2020;75(5):341-47. doi: 10.1016/j.crad.2020.03.004 [published Online First: 2020/03/29]
- Wong HYF, Lam HYS, Fong AH, et al. Frequency and Distribution of Chest Radiographic Findings in Patients Positive for COVID-19. Radiology 2020;296(2):E72-E78. doi: 10.1148/radiol.2020201160 [published Online First: 2020/03/29]
- Wu J, Wu X, Zeng W, et al. Chest CT Findings in Patients With Coronavirus Disease 2019 and Its Relationship With Clinical Features. Invest Radiol 2020;55(5):257-61. doi: 10.1097/RLI.0000000000000670 [published Online First: 2020/02/25]
- Xie X, Zhong Z, Zhao W, et al. Chest CT for Typical Coronavirus Disease 2019 (COVID-19) Pneumonia: Relationship to Negative RT-PCR Testing. Radiology 2020;296(2):E41-E45. doi: 10.1148/radiol.2020200343 [published Online First: 2020/02/13]
- Xu X, Yu C, Qu J, et al. Imaging and clinical features of patients with 2019 novel coronavirus SARS-CoV-2. Eur J Nucl Med Mol Imaging 2020;47(5):1275-80. doi: 10.1007/s00259-020-04735-9 [published Online First: 2020/02/29]
- Yang W, Cao Q, Qin L, et al. Clinical characteristics and imaging manifestations of the 2019 novel coronavirus disease (COVID-19):A multi-center study in Wenzhou city, Zhejiang, China. J Infect 2020;80(4):388-93. doi: 10.1016/j.jinf.2020.02.016 [published Online First: 2020/03/01]
- Zhang JJ, Dong X, Cao YY, et al. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020;75(7):1730-41. doi: 10.1111/all.14238 [published Online First: 2020/02/23]
- Zhao W, Zhong Z, Xie X, et al. CT Scans of Patients with 2019 Novel Coronavirus (COVID-19) Pneumonia. Theranostics 2020;10(10):4606-13. doi: 10.7150/thno.45016 [published Online First: 2020/04/16]
- Zhao W, Zhong Z, Xie X, et al. Relation Between Chest CT Findings and Clinical Conditions of Coronavirus Disease (COVID-19) Pneumonia: A Multicenter Study. AJR Am J Roentgenol 2020;214(5):1072-77. doi: 10.2214/AJR.20.22976 [published Online First: 2020/03/04]
- Zhou Y, Zhang Z, Tian J, et al. Risk factors associated with disease progression in a cohort of patients infected with the 2019 novel coronavirus. Ann Palliat Med 2020;9(2):428-36. doi: 10.21037/apm.2020.03.26 [published Online First: 2020/04/03]
- Guan WJ, Ni ZY, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med 2020;382(18):1708-20. doi: 10.1056/NEJMoa2002032 [published Online First: 2020/02/29]
- Liu W, Zhang Q, Chen J, et al. Detection of Covid-19 in Children in Early January 2020 in Wuhan, China. N Engl J Med 2020;382(14):1370-71. doi: 10.1056/NEJMc2003717 [published Online First: 2020/03/13]
- Xia W, Shao J, Guo Y, et al. Clinical and CT features in pediatric patients with COVID-19 infection: Different points from adults. Pediatr Pulmonol 2020;55(5):1169-74. doi: 10.1002/ppul.24718 [published Online First: 2020/03/07]
- Liu M, Song Z, Xiao K. High-Resolution Computed Tomography Manifestations of 5 Pediatric Patients With 2019 Novel Coronavirus. J Comput Assist Tomogr 2020;44(3):311-13. doi: 10.1097/RCT.0000000000001023 [published Online First: 2020/03/29]
- Zheng F, Liao C, Fan QH, et al. Clinical Characteristics of Children with Coronavirus Disease 2019 in Hubei, China. Curr Med Sci 2020;40(2):275-80. doi: 10.1007/s11596-020-2172-6 [published Online First: 2020/03/25]
- Lin J, Duan J, Tan T, et al. The isolation period should be longer: Lesson from a child infected with SARS-CoV-2 in Chongqing, China. Pediatr Pulmonol 2020;55(6):E6-E9. doi: 10.1002/ppul.24763 [published Online First: 2020/04/04]
- Wu Q, Xing Y, Shi L, et al. Epidemiological and Clinical Characteristics of Children with Coronavirus Disease 2019. medRxiv 2020 doi: 10.1101/2020.03.19.20027078 [published Online First: March 26, 2020]
- Tang A, Tong ZD, Wang HL, et al. Detection of Novel Coronavirus by RT-PCR in Stool Specimen from Asymptomatic Child, China. Emerg Infect Dis 2020;26(6):1337-39. doi: 10.3201/eid2606.200301 [published Online First: 2020/03/10]
- Khatami F, Saatchi M, Zadeh SST, et al. A meta-analysis of accuracy and sensitivity of chest CT and RT-PCR in COVID-19 diagnosis. Sci Rep 2020;10(1):22402. doi: 10.1038/s41598-020-80061-2 [published Online First: 2020/12/30]
- Kim JE, Kim UJ, Kim HK, et al. Predictors of viral pneumonia in patients with community-acquired pneumonia. PLoS One 2014;9(12):e114710. doi: 10.1371/journal.pone.0114710 [published Online First: 2014/12/23]
- Bai HX, Hsieh B, Xiong Z, et al. Performance of Radiologists in Differentiating COVID-19 from Non-COVID-19 Viral Pneumonia at Chest CT. Radiology 2020;296(2):E46-E54. doi: 10.1148/radiol.2020200823 [published Online First: 2020/03/11]
- Zimmermann P, Curtis N. Coronavirus Infections in Children Including COVID-19: An Overview of the Epidemiology, Clinical Features, Diagnosis, Treatment and Prevention Options in Children. Pediatr Infect Dis J 2020;39(5):355-68. doi: 10.1097/INF.0000000000002660 [published Online First: 2020/04/21]
- Lee PI, Hu YL, Chen PY, et al. Are children less susceptible to COVID-19? J Microbiol Immunol Infect 2020;53(3):371-72. doi: 10.1016/j.jmii.2020.02.011 [published Online First: 2020/03/10]

