Drug vignettes: Tocilizumab
May 1, 2020

Robin E Ferner*, Jeffrey K Aronson
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
*University of Birmingham
Correspondence to r.e.ferner@bham.ac.uk
Updated 3 August 2020, 26 October 2020, 20 January 2021, 28 July 2021
Patients with severe COVID-19 can develop cytokine release syndrome (“cytokine storm”) and are reported to have high circulating IL-6 concentrations. Tocilizumab is a recombinant humanized anti-human IgG1 monoclonal antibody directed against the interleukin-6 receptor (IL-6R). It has therefore been proposed to be of value in the management of severe COVID-19.
Mechanism of action
Tocilizumab binds specifically to both soluble and membrane-bound interleukin-6 receptor (IL-6R) with a pKd of 8.6 (i.e. Kd = 2.5 nmol/L). It is a competitive inhibitor of IL-6-mediated signalling.
IL-6 is a cytokine involved in haemopoiesis, acute phase reactions, and immune responses, such as T-cell activation and induction of immunoglobulin secretion. It is produced by T- and B-lymphocytes, monocytes, fibroblasts, and other cells. Excessive production of IL-6 is implicated in, for example, multiple myeloma and rheumatoid arthritis.
In an animal model, tocilizumab suppressed the development of arthritis and the synthesis of C-reactive protein (CRP).
Circulating IL-6 concentrations are raised in patients with SARS and COVID-19. IL-6 activates a downstream Janus kinase (JAK) and excessive signalling leads to many effects that contribute to organ damage, such as maturing naïve T cells into effector T cells, inducing expression of vascular endothelial growth factor (VEGF) in epithelial cells, increasing vascular permeability, and reducing myocardial contractility.
Indications for licensed formulations
Tocilizumab is licensed in the UK and the USA, with some limitations, for the treatment of:
- severe, active, and progressive rheumatoid arthritis;
- active systemic juvenile idiopathic arthritis;
- juvenile idiopathic polyarthritis, in combination with methotrexate (MTX);
- giant cell arteritis (subcutaneous formulation)
- chimeric antigen receptor (CAR) T cell-induced severe or life-threatening cytokine release syndrome (CRS).
All of these indications are to treat chronic diseases, apart from the cytokine release syndrome, for which the FDA approved tocilizumab in 2017, on the basis of retrospective evidence of its effectiveness observed when it was used during prospective clinical trials of other interventions. It had previously been approved for the treatment of the chronic conditions listed above.Practical use in licensed indications
Tocilizumab is marketed in the UK by Roche Products Limited as RoActemra, in solutions containing 162 mg for injection (pens and syringes) and as a 20 mg/mL concentrate from which a solution for infusion can be prepared.
The effects of tocilizumab have been studied in several masked, controlled, randomized studies in rheumatoid arthritis. In a 52-week trial of nearly 800 patients, 20% of those treated with tocilizumab (8 mg/kg every four weeks) + methotrexate achieved a major clinical response (ARC 70), compared with 4% receiving placebo + methotrexate.
In a systematic review of 41 double-blind, randomized, controlled trials, non-controlled clinical trials, and open-label extensions lasting at least 6 months in patients with rheumatoid arthritis, tocilizumab monotherapy produced greater improvement than methotrexate monotherapy, as did the combination in patients who had failed to respond to methotrexate or anti-TNF therapy. However, the rate of serious infections was 4.7 events/100 patient-years of exposure in those who were given tocilizumab and there were also higher frequencies of neutropenia, thrombocytopenia, hyperlipidemia, and transaminase rises compared with placebo.
In rheumatic disease, tocilizumab is given by intravenous infusion once every four weeks, adjusted to the patient’s weight. The maximum recommended single dose is 800 mg. It can also be given by subcutaneous injection 162 mg once a week.
The dosage in the treatment of the cytokine release syndrome is a 60-minute intravenous infusion of 8 mg/kg in patients weighing 30 kg or more and 12 mg/kg in patients weighing less than 30 kg.
Pharmacokinetics
Tocilizumab has complicated pharmacokinetics. Its clearance depends on two pathways, one linear, one nonlinear. The nonlinear clearance is rapidly saturated at low concentrations, so that at high concentrations the linear clearance predominates and is about 10 mL/h, i.e. very low (compared with liver blood flow of 80 L/h). Thus, tocilizumab has a very long and dose-dependent half-life, increasing from 6 days at low concentrations to 18 days at high concentrations. The apparent volume of distribution is about 7 L. Mean steady-state trough serum concentrations (i.e. concentration at the end of a dosing interval) are about 45 mg/L at a weekly subcutaneous dose of 162 mg and 19 mg/L during 4-weekly intravenous administration.
Contraindications and warnings in licensed use
Serious and sometimes fatal infections have been reported. The manufacturer therefore recommends that treatment should not be started during an active severe infection, or with conditions that can predispose to infections (e.g. diverticular disease, diabetes mellitus, interstitial lung disease). It is not clear how these restrictions would affect its use in acute infection with SARS-CoV-2.
Tocilizumab can also cause reactivation of tuberculosis and hepatitis B virus infection.
Hypersensitivity to tocilizumab or any of the excipients in the formulations is a contraindication. This is a typical blanket warning that is included in Summaries of Product Characteristics. However, acute hypersensitivity reactions to tocilizumab are not uncommon with repeated administration (see below).
The manufacturers advise against starting treatment in patients with a neutrophil count below 2.0 × 109/L or a platelet count below 100 × 1012/L. They advise withholding treatment if the neutrophil count falls below 0.5 × 109/L or the platelet count below 50 × 1012/L. They also advise caution in starting treatment in patients with liver transaminase activities more than 1.5 times the upper limit of normal and withholding treatment if transaminase activity rises above 3–5 times the upper limit of normal.
Adverse effects
Very common adverse effects (more than 1 in 10 people affected) include:
-
-
- Upper respiratory tract infections
- Hypercholesterolaemia
Common adverse effects (between 1 in 10 and 1 in 100 people affected) include:
-
-
- Herpes simplex, herpes zoster, cellulitis, pneumonia
- Gastritis and mouth ulcers
- Rashes
- Raised transaminase activities, hyperbilirubinaemia
- Hypertension
- Leukopenia, neutropenia, hypofibrinogenaemia
- Conjunctivitis
- Cough and dyspnoea
Hypersensitivity reactions occur in about 1.5% of patients with rheumatoid arthritis.
Drug-drug interactions
During cytokine storm drug metabolism by CYP isoenzymes is suppressed. Thus, successful treatment with tocilizumab may return to normal the metabolism of other drugs, such as methylprednisolone and dexamethasone.
Experience with tocilizumab in cytokine release syndrome
Administration of chimeric antigen receptor T (CAR-T) cells can cause a cytokine release syndrome, which has been treated with tocilizumab. The appropriate timing of therapy has not been well described.
The IL-6 concentration in 69 patients correlated with concentrations of CRP, ferritin, and D-dimers. An improvement in the radiographic appearance was accompanied by reduction in IL-6 concentration. However, since IL-6 clearance is mediated by the IL-6 receptor, blocked by tocilizumab, IL-6 accumulates in the serum during tocilizumab treatment, so it may not be a good marker of disease activity in tocilizumab-treated patients.
Experience with tocilizumab in virus infections other than COVID-19
IL-6 generated in damaged lung tissue has been implicated in the inflammatory myopathy that can occur in influenza A, and tocilizumab attenuated influenza-induced muscle dysfunction in experiments in mice.
The authors of a Japanese study of the course of influenza A infection in ten patients with juvenile rheumatoid arthritis receiving tocilizumab treatment concluded that “IL-6 inhibition by tocilizumab reduced inflammation associated with infection and resulted in mild symptoms during influenza.” Eight of the ten patients also received oseltamivir or zanamivir.
Experience with tocilizumab in COVID-19
Typical protocols include:
-
-
- 400 mg intravenously;
- 8 mg/kg on day 1, repeated on day 3 if there is no response;
- 8 mg/kg up to a maximum of 800 mg, repeated once if symptoms worsen or there is no response.
Non-randomized observations
Cao noted a small, apparently uncontrolled, Chinese trial of tocilizumab in which 21 patients with severe disease, including two who were critically ill, recovered. No other clinical data were provided.
A French patient with metastatic renal cell cancer developed fever, tested positive for SARS-CoV-2, and was treated with lopinavir–ritonavir. His condition deteriorated suddenly on day 8 of his illness, he was given two intravenous doses of tocilizumab 8 mg/kg six hours apart, and his clinical state improved rapidly.
A Chinese group reported the effects of tocilizumab in 15 patients, of whom seven were said to be critically ill. Eight patients were treated with methylprednisolone in addition. “Although TCZ treatment ameliorated the increased CRP in all patients rapidly, for the 4 critically ill patients who received only single dose of TCZ, 3 of them (No. 1, 2, and 3) still dead [sic] and the CRP level in the rest 1 patient (No. 7) failed to return to normal range with a clinical outcome of disease aggravation.” The authors reasonably concluded that, while a single dose of tocilizumab in combination with methylprednisolone failed to reduce disease activity, there was some suggestion that repeated doses of tocilizumab were clinically beneficial.
The effects of tocilizumab in addition to routine therapy have been retrospectively analysed in 21 Chinese patients with severe or critical COVID-19. The authors of a non-peer-reviewed account of the results, subsequently published, reported that within a few days fever abated and all other symptoms improved; 15 patients needed less oxygen intake and one needed none at all; lung opacities in CT scans “were absorbed” in 19 patients. The peripheral lymphocyte counts, which were below normal in 17 of 20 patients before treatment, returned to normal in 10 out of 19 by 5 days, and abnormally high C-reactive protein concentrations fell significantly in 16 out of 19 patients. These are apparently encouraging results, but the study was small and retrospective and there was no control group for comparison. In different parts of the paper the authors refer to either 20 or 21 patients; in the tables, the demographic data refer to 21, but the laboratory data refer to anything from 12 to 20 patients or do not specify; the results of oxygen therapy are described in 20 patients. We have not found a prespecified protocol.
Randomized studies
On 29 July 2020 Roche released a statement on the randomized, masked, placebo-controlled COVACTA trial of tocilizumab in patients with severe COVID-19, titled “Roche provides an update on the phase III COVACTA trial of Actemra/RoActemra [tocilizumab] in hospitalised patients with severe COVID-19 associated pneumonia”.
The trial protocol, titled “A Study to Evaluate the Safety and Efficacy of Tocilizumab in Patients With Severe COVID-19 Pneumonia”, indicated that the intention was to recruit 450 patients, but the company statement does not give data on how many patients were recruited or how they were distributed between the active and placebo arms.
The specified primary endpoint was “Clinical Status Assessed Using a 7-Category Ordinal Scale [Time Frame: Day 28]”. The results posted on 29 July stated that the odds ratio for clinical improvement at four weeks did not differ significantly between treatments: odds ratio in favour of tocilizumab = 1.19 (95% Confidence Interval 0.81–1.76), and that mortality at 4 weeks was 19.7% in the tocilizumab and 19.4% in the placebo group (P = 0.9410).
Other measures included:
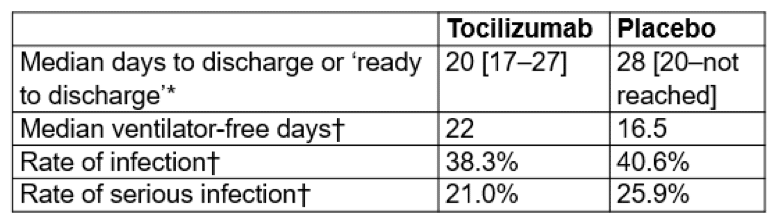
* P=0.0370. “However, the difference cannot be considered statistically significant as the primary endpoint was not met.” Nor was any correction made for multiple comparisons.
†P greater than 0.05
- “COVACTA trial did not meet its primary endpoint of improved clinical status in patients with COVID-19 associated pneumonia, or the key secondary endpoint of reduced patient mortality
- The study is the first global, randomised, double-blind, placebo-controlled phase III trial investigating [tocilizumab] in this setting”
They also stated that “In addition, the key secondary endpoints, which included the difference in patient mortality at week four, were not met; however, there was a positive trend in time to hospital discharge in patients treated with [tocilizumab]. The COVACTA study did not identify any new safety signals for [tocilizumab].”
These results do not change our conclusion, described under “Outlook” below.
Further evidence (added on 26 October 2020)
The results of a study of the effects of tocilizumab in patients with confirmed SARS-CoV-2 infection and evidence of systemic inflammation have now been published. The Genentech-sponsored study was a randomized, double-blind, placebo-controlled trial conducted in seven hospitals in Boston, Massachusetts. The protocol was pre-published.
The patients were adults with symptomatic SARS-CoV-2 infection confirmed either by PCR or serum IgM assay, and who had an inflammatory response as demonstrated by one or more of:
serum C-reactive protein concentration above 50 mg/L;
serum ferritin concentration above 500 mg/L;
serum D-dimer concentration above 1 microgram/L;
serum lactate dehydrogenase activity above 250 units/L,
provided they were not so severely ill that they required high-flow oxygen or ventilatory assistance.
Treatment was standard care plus tocilizumab by infusion 8 mg/kg body weight up to a maximum of 800 mg, or placebo. Of 1560 patients who were screened, 243 were randomized; of those, 242 were treated with tocilizumab (161 patients) or placebo (81 patients). In the tocilizumab arm there were more patients over the age of 65 years, more men, and more white people; in the placebo arm there were more patients with diabetes.
The primary outcome measure was death or mechanical ventilation by 28 days from enrolment in the trial. Several subsidiary outcomes, including death alone and mechanical ventilation alone, were also examined.
Of adverse events, neutropenia was more common in the tocilizumab group. Selected results are shown in the Table.
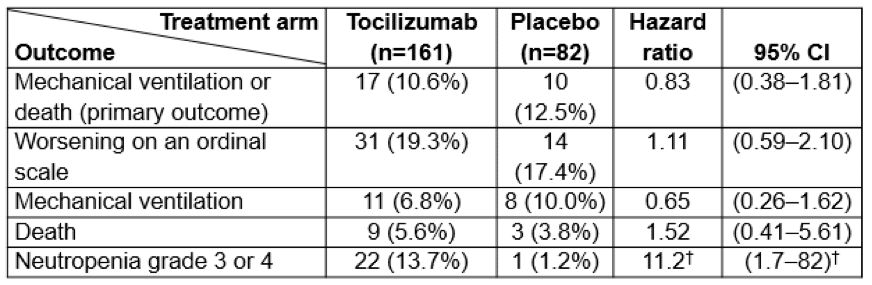
†Calculated by us
The authors concluded that “Our data do not provide support for the concept that early interleukin-6 receptor blockade is an effective treatment strategy in moderately ill patients hospitalized with Covid-19.” We agree. We also agree that “The overall experience with interleukin-6 receptor blockade, however, underscores the point that any such approach must be subjected to randomized, blinded trials. Moreover, such investigations should be performed early in the course of evaluating each strategy, before the adoption of widespread use.”
Further evidence (added on 13 January 2021)
Further details of the REMAP-CAP results were published in preliminary form on 7 January 2021, and some information from the tocilizumab arm of RECOVERY was published on 8 January 2021.
REMAP-CAP used as a primary endpoint an ordinal scale that combined death in hospital (assigned a score of –1) and days free from organ support up to day 21. This was markedly different from the primary endpoint of RECOVERY, which was mortality at 28 days.
A Bayesian analysis of REMAP-CAP showed that both tocilizumab and sarilumab, another anti-IL-6 receptor monoclonal antibody, were significantly better than usual care.
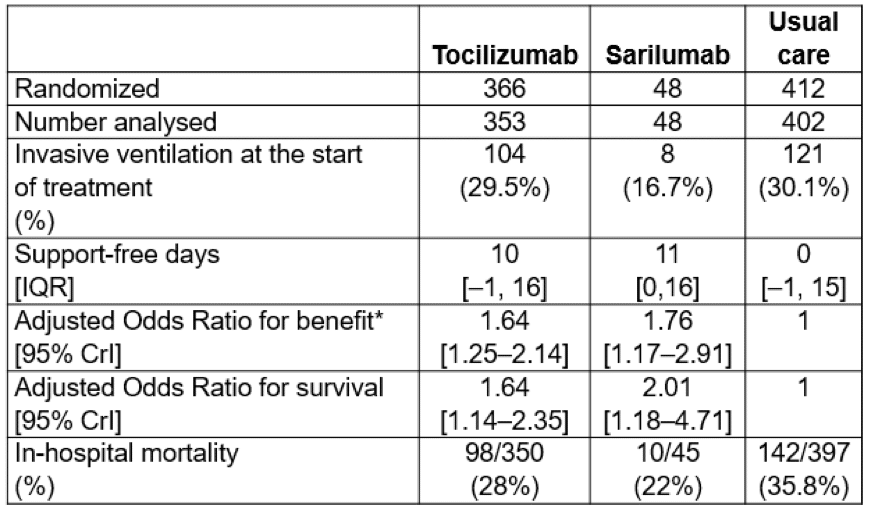
* Improved survival and/or more organ support-free days
At least 651/812 patients had received dexamethasone or hydrocortisone, and 265/807 received remdesivir
The RECOVERY investigators also reported a meta-analysis of six published randomized trials (Table 1), in which 1223 patients were given an anti-IL-6 receptor monoclonal antibody, mostly tocilizumab but alternatively, in some cases in REMAP-CAP, sarilumab; 884 were given some form of usual care. Treatment with an anti-IL-6 receptor antibody non-significantly reduced the odds of dying compared with usual care: absolute risk reduction 4.6%, Odds Ratio 0.83 (0.66–1.04), P=0.11. The result was dominated by the outcome in the REMAP-CAP trial. Neither adverse events nor any measures of heterogeneity were reported in the meta-analysis. Nor have any direct comparisons been made between the use of anti-IL-6 receptor antibodies with or without the use of a corticosteroid, such as dexamethasone, since the combination might be more effective than either used alone.
In contrast, the RECOVERY investigators have randomized over 2900 participants to tocilizumab or usual care, without detecting a clear signal that it benefits survival.
Table 1. Some brief details of six randomized trials of anti-IL-6 receptor antibodies in covid-19
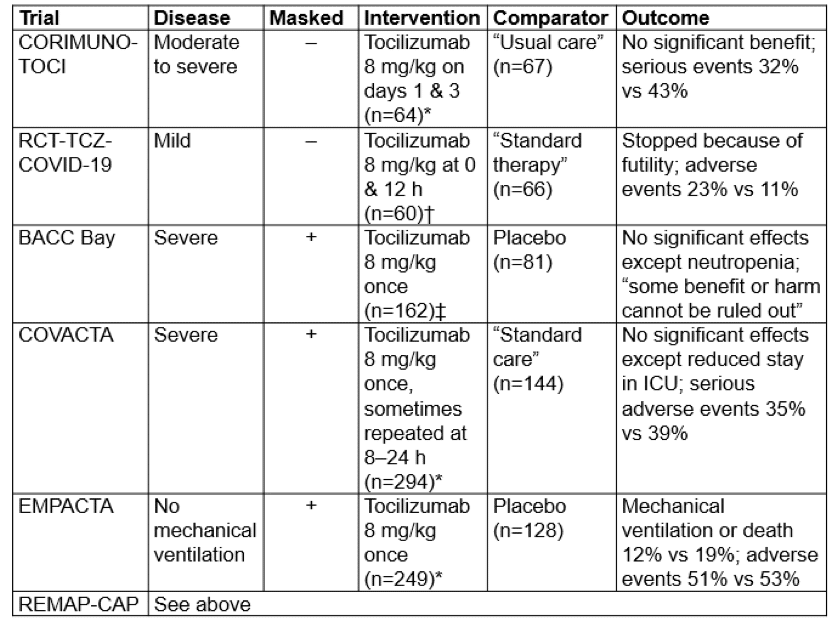
*Corticosteroids could be used
†Corticosteroids were used only if they had already been in use before admission
‡”glucocorticoids were permitted” but “no patients received dexamethasone”
Links: CORIMUNO-TOCI; RCT-TCZ-COVID-19; BACC Bay; COVACTA; EMPACTA; REMAP-CAP
Yet more evidence (added on 23 January 2021)
An open-label randomized trial in nine Brazilian hospitals comparing a single infusion of 8 mg/kg tocilizumab + standard care with standard care alone in 129 patients with moderate to severe COVID-19 was reported on 20 January 2021. Efficacy was assessed using an intention-to-treat analysis (two patients in the control arm received tocilizumab). The protocol was prepublished (NCT04403685).
The trial population consisted of patients with PCR-positive SARS-CoV-2 infection and raised inflammatory markers who had been symptomatic for more than three days and who required supplemental oxygen to maintain SaO2 above 93%. Patients ventilated for more than 24 hours and those with active uncontrolled infection other than COVID-19, liver disease, or kidney disease were excluded, as were pregnant or breastfeeding women.
The primary outcome was status on a 7-point ordinal scale at 15 days after trial entry. Secondary outcomes included mortality at 28 days and days free from mechanical ventilation up to 29 days after trial entry. The binary outcomes “alive, not receiving mechanical ventilation” (status 1–5) and “receiving mechanical ventilation or dead” (status 6 or 7) were assessed by logistic regression.
The trial was halted before the intended 150 patients were recruited because an interim analysis showed excess mortality in the tocilizumab arm.
The groups were similar at baseline and received similar non-trial treatments, except that at baseline fewer patients in the tocilizumab group received high-flow oxygen or non-invasive or invasive ventilation (19% versus 57%) and fewer received azithromycin (35% versus 48%).
During the first 15 days, 10% of the tocilizumab group and 4.8% of the control group received an antiviral drug, while 84% and 89% respectively received a corticosteroid.
Other results were:
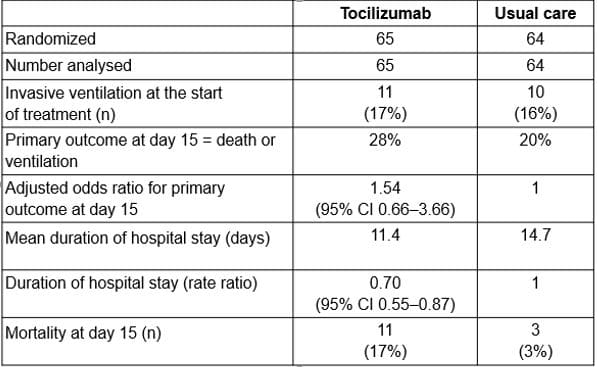
This small open randomized trial failed to show a survival benefit and was halted after 86% of target recruitment. The duration of hospital stay was reduced, but there was a worse overall outcome and numerically higher mortality at 15 days in those treated with tocilizumab.
Outlook
Current evidence from randomized clinical trials on the efficacy of anti-IL-6 receptor antibodies in covid-19 is contradictory, and although it appears that overall there may be a small reduction in mortality, the results are not convincing. However, the reduction in duration of respiratory and cardiac support found in REMAP-CAP, and the pressure on intensive care beds in the NHS has encouraged the use of tocilizumab. The RECOVERY investigators will continue to recruit patients to the tocilizumab arm of the trial, and until their results become available, it would be preferable if eligible patients were recruited to the randomized trial.
Registered trials
When last (27 April 2020) we surveyed trials involving tocilizumab, either alone or in combination with other medications, we found 35. Now, in the WHO’s list dated 14 January 2021, we have found 90 studies, of which eight are observational studies. Of the 82 registered clinical trials 61 are randomized, but only 16 are masked (one single-masked, investigator). In all, 4703 participants are scheduled to be studied in the masked trials compared with 31,202 in the unmasked trials (15%).
Emergency Use Authorization by the FDA announced on 24 June 2021
The US Food & Drug Administration has now issued an Emergency Use Authorization for the Actemra® brand of tocilizumab, for treatment of hospitalized adults and children aged 2 years and older, who are receiving systemic corticosteroids and require supplementary oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). This is based on evidence from four clinical trials that tocilizumab reduces the risk of death during 28 days of follow-up and reduces the amount of time patients spend in hospital.
Actemra-tocilizumab is not authorized for use in outpatients with COVID-19.
Genentech Inc have provided a Fact Sheet, which includes the following data on mortality at 28 days:
Thus, 809 deaths occurred within 28 days of randomization in 2576 patients who received usual care and 783 deaths in 2995 tocilizumab-treated patients, that is 31.4% versus 26.1% in favour of tocilizumab. Benefit in RECOVERY, the largest trial, was seen in those who received both tocilizumab and dexamethasone. The hazard ratio among the pre-specified subgroup of patients who received systemic corticosteroids at baseline was 0.79 (95% CI: 0.70 to 0.89), and in the pre-specified subgroup who did not receive systemic corticosteroids at baseline the hazard ratio was 1.16 (95% CI: 0.91 to 1.48).
Neither the FDA nor Genentech’s Fact Sheet included results from other studies, including the REMAP-CAP study of tocilizumab and sarilumab, in which in-hospital mortality was reduced from 142/397 (36%) in the control group to 98/350 (28%) by tocilizumab and 10/45 (22%) by sarilumab. In that study, 93-95% of the patients in the three groups also received a corticosteroid within 48 hours of randomization.
Disclaimer: This article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.

