Drug vignettes: Dexamethasone
June 26, 2020
Robin E Ferner*†, Nicholas DeVito, Jeffrey K Aronson
On behalf of the Oxford COVID-19 Evidence Service Team
Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences
University of Oxford
*University of Birmingham
†University College London
Correspondence to r.e.ferner@bham.ac.uk
Description of the drug
Dexamethasone (9α-fluoro-16α-methylprednisolone) is a potent corticosteroid with predominantly glucocorticoid effects. It has almost no mineralocorticoid action. It was first synthesized in 1957, in an attempt to find a steroid with a longer duration of action than hydrocortisone and other steroids available at that time. This was done by the addition of a methyl group on the cyclopentane ring (Figure 1).
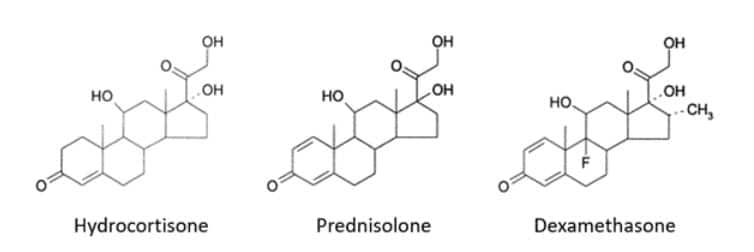
Figure 1. Structures of some corticosteroids
The addition of a fluorine atom in the 9α position made the compound more potent as a glucocorticoid. Because of differences in oral availability, 1 mg of dexamethasone by mouth has the glucocorticoid activity of about 8 mg of prednisolone or 25 mg of hydrocortisone.
Mechanism of action
Corticosteroids are endogenous hormones produced in the adrenal cortex or their synthetic analogues. Glucocorticoids enter cells and bind to intracellular receptors. Hydrolysis of an attached heat-shock protein releases the activated steroid-receptor complex, which is transported into the nucleus, where it binds to DNA and modifies DNA glucocorticoid-responsive elements and transcription factors. The modifications alter transcription of target genes in DNA into messenger RNA; mRNA leaves the nucleus and directs the synthesis of new proteins on cytoplasmic ribosomes; these proteins are released from the cell and elicit biological responses.
Glucocorticoids in low and high doses increase the production of anti-inflammatory compounds, such as annexin-1, SLP1, MOP-1, IκB-α, GILZ, and nitric oxide synthase, and in high doses reduce the production of pro-inflammatory compounds, including cytokines, chemokines, adhesion molecules, and pro-inflammatory enzymes, such as phospholipase A2 and cyclooxygenase.
The consequences of glucocorticoid actions are widespread, and include: mobilization of glucose through increased gluconeogenesis, causing hyperglycaemia, and thus increased insulin secretion and increased glycogen storage; redistribution of body fat; breakdown of protein; neutrophilia and lymphopenia; and immunosuppression. The immunosuppression arises partly from inhibition of kinases responsible for cytokine production and partly from inhibition of the nuclear transcription factor NF-κB, which stimulates the transcription of cytokines, chemokines, and other molecules in the inflammatory pathway.
Glucocorticoids also activate histone deacetylase (HDAC2) by activating transcription factors such as CREB, AP-1, and NF-κB. This activates chromatin, which increases gene transcription.
Glucocorticoids increase apoptosis in inflammatory cells such as eosinophils, T lymphocytes, mast cells, and macrophages, reducing cellular immune responses and production of cytokines.
Proposed mechanism of action in COVID-19
The sickest patients with COVID-19 suffer a hyperinflammatory state—a cytokine storm—that has features in common with a rare haematological condition called haemophagocytic lymphohistiocytosis. Immune suppression should help such patients. By contrast, immune suppression during the early phase of the viral infection might allow increased viral replication and aggravate the disease.
The 3C-like proteinase on SARS-CoV-2 (nsp5) inhibits HDAC2 transport into the nucleus, and so impairs the way in which it mediates inflammation and cytokine responses, so activation of histone deacetylase by dexamethasone may directly oppose the action of SARS-CoV-2.
Practical use in licensed indications
The Electronic Medicines Compendium lists 35 formulations of dexamethasone: tablets and oral solutions (17 formulations), solutions for injection (6 formulations), an ointment (1 formulation), ear drops or spray and eye drops (10 formulations), and an intravitreal implant. The strengths of the formulations range from 0.5 to 4 mg per tablet and 2 to 20 mg/5mL of oral solution.
Under the heading “Therapeutic indications” the Summaries of Product Characteristics state that dexamethasone (2 mg tablets) is “Indicated in a wide variety of disorders amenable to glucocorticoid therapy, as well as an adjunct in the control of cerebral oedema.”
The solution for injection is indicated “For use in certain endocrine and non-endocrine disorders responsive to corticosteroid therapy.”
Thus, the indications for glucocorticoids in therapeutic practice are very wide, and practical use varies accordingly. For example, a dose sufficient to suppress cortisol (hydrocortisone) secretion by the adrenal gland in healthy subjects, used in the dexamethasone suppression test, is dexamethasone 1 mg by mouth. A dose of 16 mg daily is used in patients with brain tumours to relieve the symptoms of raised intracranial pressure. The oral dosage range for treating inflammatory and allergic disorders in adults is 0.5–10 mg/day.
Pharmacokinetics
In a study of dexamethasone kinetics in two groups of 15 patients with community-acquired pneumonia, one group was treated with dexamethasone 6 mg/day by mouth and the other with 4 mg/day intravenously. The apparent volume of distribution was 1 L/kg in both groups, but the half-life after oral administration was approximately 7 hours, and after intravenous administration 9 hours. The bioavailability of oral dexamethasone was 81% (95% CI = 54–121%).
However, the biological half-life of dexamethasone is much longer, of the order of 36–54 h compared with 18–36 h for prednisolone.
Known contraindications and cautions
Formal contraindications are: “systemic fungal infection; systemic infection, unless specific anti-infective therapy is employed; hypersensitivity to the active ingredient or any other component of the medication; administration of live virus vaccines.”
The British National Formulary includes an extensive list of cautions, many related to the known effects of corticosteroids (see above). Conditions in which caution is required in short-term use, as in COVID-19, include:
- congestive heart failure
- diabetes mellitus (including a family history)
- diverticulitis
- epilepsy
- glaucoma (including a family history or susceptibility)
- history of steroid myopathy
- history of tuberculosis or X-ray changes (frequent monitoring required)
- hypertension
- hypothyroidism
- infection (particularly untreated)
- myasthenia gravis
- ocular herpes simplex (risk of corneal perforation)
- osteoporosis (post-menopausal women and the elderly at risk)
- (in adults) peptic ulceration
- recent intestinal anastomoses
- recent myocardial infarction (rupture reported)
- thromboembolic disorders
- ulcerative colitis.
In addition:
- psychiatric adverse effects can occur, especially in those with an existing or previous history of severe affective disorders in themselves or in their first-degree relatives
- susceptibility to infection is increased, and signs of infection can be masked
- chickenpox and measles can be severe or fatal in patients taking immunosuppressive doses of corticosteroids.
Because dexamethasone is used only for short-term treatment in COVID-19, many of these cautions may not apply. However, we do not know whether dexamethasone increases the risk of thromboembolic disorders in acute COVID-19 infection.
Known and potential adverse effects
Important adverse effects of systemic glucocorticoids include adrenal suppression, susceptibility to and reactivation of infection, masking of clinical signs of infection, disturbed carbohydrate metabolism and diabetes, hypertension, heart failure, proximal muscle weakness, glaucoma, cataract, and avascular necrosis of bone. Disturbed electrolyte balance (e.g. hypokalaemia) is most likely with corticosteroids that have significant mineralocorticoid activity (e.g. hydrocortisone); it is generally not a problem with dexamethasone.
Most of these effects are problematic during long-term rather than short-term therapy, although hyperglycaemia may have to be managed during short-term treatment.
Drug–drug interactions
Many potentially serious drug–drug interactions with corticosteroids in general, and with dexamethasone specifically, are recognized. Dexamethasone increases the risk of peptic ulceration and bleeding if taken with non-steroidal anti-inflammatory drugs and of gastrointestinal ulceration with nicorandil. However, non-steroidal anti-inflammatory drugs should not be used in patients with severe COVID-19 and nicorandil is unlikely to be.
Dexamethasone can cause hypokalaemia, and so potentiate the risk of ventricular arrhythmias if it is given with drugs that prolong the QT interval; however, it has little mineralocorticoid activity, so the risk is small. This might be important in patients who are being given azithromycin or hydroxychloroquine, drugs that have been advocated for treatment of COVID-19, although we do not believe that there is any benefit in using hydroxychloroquine.
Dexamethasone increases the risk of severe systemic infection with live vaccines.
Experience in other viral infections
There is little evidence of benefit from glucocorticoids in general or dexamethasone in particular in viral infections other than COVID-19.
Meta-analysis of studies in children with acute viral bronchiolitis shows no significant improvement with glucocorticoids.
A randomized controlled trial in 65 patients found no benefit in Japanese encephalitis virus infection.
An old report of two cases suggested that dexamethasone might be beneficial in cases of herpes simplex encephalitis; but as correspondents pointed out at the time: “It is true that clinical experience has not always borne out the dire forebodings of virologists, yet it is also true that, with particular reference to herpes-simplex virus infection, the exhibition of steroids has often led to a worsening of the disease and occasionally disastrous results”; there is no general enthusiasm for its use in this indication.
The authors of a systematic review of the effects of corticosteroids in dengue fever concluded that “the evidence from trials using corticosteroids in dengue is inconclusive and the quality of evidence is low to very low.”
A retrospective study of 401 patients with Severe Acute Respiratory Syndrome (SARS) from Coronavirus-1 suggested that corticosteroid use might reduce the mortality in “critical” cases.
A retrospective cohort study of early corticosteroid treatment in 241 patients with adult respiratory distress syndrome (ARDS) associated with influenza infection showed a significant increase in hospital mortality: 37/85 patients (44%) in the corticosteroid cohort died, but only 30/156 (19%) of the control patients—adjusted odds ratio = 5.02 (95% CI = 2.39–10.54; P < 0.001). Even though the authors of this study corrected as far as possible for factors that might have influenced the outcome, retrospective studies are inherently unreliable.
In adult respiratory distress syndrome of any cause, a prospective randomized controlled trial of methylprednisolone in 180 patients failed to show benefit, although an intention-to-treat analysis of the same data subsequently sought to show that the lack of benefit was a consequence of rapid discontinuation of the glucocorticoid.
Early administration of dexamethasone in an open randomized controlled trial of 277 patients who required mechanical ventilation for ARDS reduced overall mortality. In the dexamethasone group, hospital mortality was 33/139 (24%), compared with 50/138 (36%) in the control group, a risk difference of –12.5%, relative risk 0.66 (95% CI = 0.45–0.95; P=0.02). However, the trial did not distinguish between different causes of ARDS, which included sepsis, aspiration, trauma, and pneumonia, so its relevance to COVID-19 is unclear.
A recent meta-analysis of 10 trials concluded that the risk of dying was reduced by prolonged corticosteroid therapy started before day 14 of ARDS, relative risk 0.67 (95% CI = 0.52–0.87), but again, the causes of ARDS were undifferentiated.
Experience in COVID-19
Shang et al published the views of Chinese experts who had treated COVID-19 in Wuhan. They stated that “systemic corticosteroids should probably not be used for the treatment of COVID-19. For critically ill patients with ARDS at an early stage, corticosteroids should probably be prudently used at a low or moderate dose over the short course if there are no contraindications (Grade 2–, weak recommendation).”
There was weak supporting evidence from non-randomized trials. A cohort study of 201 patients from Wuhan showed that in the 84 patients who developed ARDS (42%), “treatment with methylprednisolone decreased the risk of death (HR, 0.38; 95% CI, 0.20–0.72).” A retrospective assessment of 46 severely ill Chinese patients showed that COVID-19 resolved more rapidly in the 26 patients given low-dose methylprednisolone. A US retrospective examination of data from patients treated with methylprednisolone and historical controls suggested that active treatment halved mortality. A similar French study, with propensity score matching, gave similar results.
We now have preliminary results from the dexamethasone arm of the UK RECOVERY Trial. The NHS Central Alerting System posted a letter from the UK’s four Chief Medical Officers and the Director of NHS England, providing scanty details: “Dexamethasone 6 mg once per day (either by mouth or by intravenous injection) for ten days was compared with 4321 UK patients randomised to usual care alone. Dexamethasone reduced deaths by one-third in ventilated patients (rate ratio 0.65 [95% confidence interval 0.48 to 0.88]; P=0.0003) and by one fifth in other patients receiving oxygen only (0.80 [0.67 to 0.96]; P=0.0021). There was no benefit among those patients who did not require respiratory support (1.22 [0.86 to 1.75; P=0.14).” The Press Release from the investigators clarified one of the problems with this letter, making it clear that 2104 patients had been randomized to dexamethasone treatment.
The data were then published in the RECOVERY investigators’ preprint mounted on medRxiv, typically marked “not certified by peer review”, on 22 June 2020. The primary endpoint was 28-day mortality. The analysis (Table 1) showed that dexamethasone reduced mortality significantly, both overall and in those receiving oxygen treatment only or invasive ventilation. However, the relative risk of dying at 28 days was non-significantly increased in those with mild disease who did not require oxygen: relative risk 1.22 (95% CI 0.93–1.61), and there was a significant trend in relative risk reduction across the three categories of severity: χ2=11.49; P<0.001. This implies that dexamethasone treatment is life-saving in patients with COVID-19 severe enough to need oxygen therapy, with or without ventilatory support, but may be harmful in patients with mild disease.
Table 1. Data taken from Table 2 of the RECOVERY investigators’ preprint in medRxiv
| |
Dexamethasone |
Control (usual care) |
RR |
95% CI |
| No oxygen received |
85/501 |
17.0% |
137/1034 |
13.2% |
1.22 |
0.93–1.61 |
| Oxygen only |
275/1279 |
21.5% |
650/2604 |
25.0% |
0.80 |
0.70–0.92 |
| Invasive mechanical ventilation |
94/324 |
29.0% |
278/683 |
40.7% |
0.65 |
0.51–0.82 |
| All participants |
454/2104 |
21.6% |
1065/4321 |
24.6% |
0.83 |
0.74–0.92
P<0.001 |
The UK’s Chief Medical Officers also wrote, “Normally we would advise waiting for the full paper before changing practice, to ensure final analysis and peer review do not lead to different conclusions. However, given this clear mortality advantage, with good significance [sic], and with a well known medicine which is safe [sic] under these circumstances we consider it is reasonable for practice to change in advance of the final paper.”
However, apart from the concern about the outcome in those who did not receive oxygen, other questions remain, such as the effects of co-medications (e.g. NSAIDs) in the different groups.
Registered clinical trials
We have identified 49 trials involving the use of corticosteroids in the treatment of COVID-19 currently listed in a variety of registries; 30 are listed in ClinicalTrials.gov, eight in the Iranian Registry of Clinical Trials, four in the EU Clinical Trials Register, three in the Chinese Clinical Trials Registry, two in the Japanese Registry of Clinical Trials, one in the Netherlands National Trial Register, and one in the Indian Clinical Trials Registry. Of these, 18 are not currently recruiting.
Only nine are double-masked and three are single-masked (investigator or outcomes assessor). These 12 studies intend to randomize 2636 participants out of a total of 22,306, i.e only 12%.
All but four are interventional studies, the others observational. Most of the former use standard care as a comparator and only 10 include a placebo; seven involve active comparators, such as tocilizumab, tacrolimus, levamisole, or siltuximab.
Most of the published protocols specify the corticosteroid to be used. In 45 cases it is a single corticosteroid: methylprednisolone (22), dexamethasone (6), ciclesonide (6), prednisone or prednisolone (5), budesonide (5), or hydrocortisone (1). In four cases more than one steroid is to be studied.
Outlook
If the full published data from the RECOVERY trial of dexamethasone in the treatment of hospitalized patients with COVID-19 confirm the results in the preliminary publications, the decision to adopt it in the NHS before publication of the final paper will have been justified.
The results in the patients who did not need oxygen seem to show no beneficial effect. However, it is not clear that in those individuals mortality is not increased by an average of 22% and perhaps up to 61%. More of the area under the probability curve certainly lies towards harm rather than benefit, although not more than 97.5%, as needed for conventional statistical significance. That being so, it will be important to avoid using dexamethasone in patients with early and mild disease and restrict it to those in the more severe categories.
There is no good evidence currently to say whether the use of corticosteroids in those who are taking them either orally for chronic inflammatory or allergic disorders or by inhalation for prevention of acute attacks of asthma will be beneficial or harmful in those exposed to, or infected by, SARS-CoV-2. People taking long-term oral corticosteroids should not stop using them, since there is a risk of an acute withdrawal syndrome (an Addisonian crisis). Those using long-term preventive inhaled corticosteroids for asthma should also continue taking them if they are deriving preventive benefit. If they are in doubt, they should seek medical advice.
Disclaimer: This article has not been peer-reviewed; it should not replace individual clinical judgement and the sources cited should be checked. The views expressed in this commentary represent the views of the authors and not necessarily those of the host institution, the NHS, the NIHR, or the Department of Health and Social Care. The views are not a substitute for professional medical advice.

